Defects in G-Actin Incorporation into Filaments in Myoblasts Derived from Dysferlinopathy Patients Are Restored by Dysferlin C2 Domains
- PMID: 31861684
- PMCID: PMC6981584
- DOI: 10.3390/ijms21010037
Defects in G-Actin Incorporation into Filaments in Myoblasts Derived from Dysferlinopathy Patients Are Restored by Dysferlin C2 Domains
Abstract
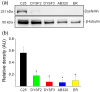
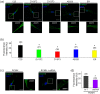
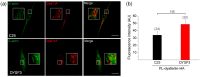
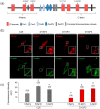
Dysferlin is a transmembrane C-2 domain-containing protein involved in vesicle trafficking and membrane remodeling in skeletal muscle cells. However, the mechanism by which dysferlin regulates these cellular processes remains unclear. Since actin dynamics is critical for vesicle trafficking and membrane remodeling, we studied the role of dysferlin in Ca2+-induced G-actin incorporation into filaments in four different immortalized myoblast cell lines (DYSF2, DYSF3, AB320, and ER) derived from patients harboring mutations in the dysferlin gene. As compared with immortalized myoblasts obtained from a control subject, dysferlin expression and G-actin incorporation were significantly decreased in myoblasts from dysferlinopathy patients. Stable knockdown of dysferlin with specific shRNA in control myoblasts also significantly reduced G-actin incorporation. The impaired G-actin incorporation was restored by the expression of full-length dysferlin as well as dysferlin N-terminal or C-terminal regions, both of which contain three C2 domains. DYSF3 myoblasts also exhibited altered distribution of annexin A2, a dysferlin partner involved in actin remodeling. However, dysferlin N-terminal and C-terminal regions appeared to not fully restore such annexin A2 mislocation. Then, our results suggest that dysferlin regulates actin remodeling by a mechanism that does to not involve annexin A2.
Keywords: C2 domains; actin; annexin A2; dysferlin; dysferlinopathy.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Structure-Based Designed Nano-Dysferlin Significantly Improves Dysferlinopathy in BLA/J Mice.Mol Ther. 2017 Sep 6;25(9):2150-2162. doi: 10.1016/j.ymthe.2017.05.013. Epub 2017 Jun 16. Mol Ther. 2017. PMID: 28629822 Free PMC article.
-
AMPK Complex Activation Promotes Sarcolemmal Repair in Dysferlinopathy.Mol Ther. 2020 Apr 8;28(4):1133-1153. doi: 10.1016/j.ymthe.2020.02.006. Epub 2020 Feb 12. Mol Ther. 2020. PMID: 32087766 Free PMC article.
-
The extracellular matrix differentially directs myoblast motility and differentiation in distinct forms of muscular dystrophy: Dystrophic matrices alter myoblast motility.Matrix Biol. 2024 May;129:44-58. doi: 10.1016/j.matbio.2024.04.001. Epub 2024 Apr 4. Matrix Biol. 2024. PMID: 38582404 Free PMC article.
-
Dysferlin function in skeletal muscle: Possible pathological mechanisms and therapeutical targets in dysferlinopathies.Exp Neurol. 2016 Sep;283(Pt A):246-54. doi: 10.1016/j.expneurol.2016.06.026. Epub 2016 Jun 25. Exp Neurol. 2016. PMID: 27349407 Review.
-
Muscle Cells Fix Breaches by Orchestrating a Membrane Repair Ballet.J Neuromuscul Dis. 2018;5(1):21-28. doi: 10.3233/JND-170251. J Neuromuscul Dis. 2018. PMID: 29480214 Free PMC article. Review.
Cited by
-
Gain-of-Function Dynamin-2 Mutations Linked to Centronuclear Myopathy Impair Ca2+-Induced Exocytosis in Human Myoblasts.Int J Mol Sci. 2022 Sep 8;23(18):10363. doi: 10.3390/ijms231810363. Int J Mol Sci. 2022. PMID: 36142275 Free PMC article.
-
Editorial of Special Issue "Frontiers in the Actin Cytoskeleton".Int J Mol Sci. 2020 May 30;21(11):3945. doi: 10.3390/ijms21113945. Int J Mol Sci. 2020. PMID: 32486337 Free PMC article.
-
N-Acetylcysteine Reduces Skeletal Muscles Oxidative Stress and Improves Grip Strength in Dysferlin-Deficient Bla/J Mice.Int J Mol Sci. 2020 Jun 16;21(12):4293. doi: 10.3390/ijms21124293. Int J Mol Sci. 2020. PMID: 32560255 Free PMC article.
-
Oxidative Stress, Inflammation and Connexin Hemichannels in Muscular Dystrophies.Biomedicines. 2022 Feb 21;10(2):507. doi: 10.3390/biomedicines10020507. Biomedicines. 2022. PMID: 35203715 Free PMC article. Review.
-
Analysis of Dysferlin Direct Interactions with Putative Repair Proteins Links Apoptotic Signaling to Ca2+ Elevation via PDCD6 and FKBP8.Int J Mol Sci. 2023 Feb 28;24(5):4707. doi: 10.3390/ijms24054707. Int J Mol Sci. 2023. PMID: 36902136 Free PMC article.
References
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

