Antimicrobial activity of resveratrol-derived monomers and dimers against foodborne pathogens
- PMID: 31862939
- PMCID: PMC6925292
- DOI: 10.1038/s41598-019-55975-1
Antimicrobial activity of resveratrol-derived monomers and dimers against foodborne pathogens
Abstract
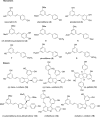
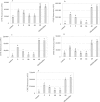
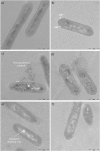
Plant polyphenolic compounds are considered a promising source for new antibacterial agents. In this study, we evaluated the antimicrobial activity of a collection of resveratrol-derived monomers and dimers screened as single molecules against a panel of nine foodborne pathogens. The results demonstrated that two monomers (i.e., pterostilbene 2 and (E)-3-hydroxy-4',5-dimethoxystilbene 9) and three dimers (i.e., δ-viniferin 10, viniferifuran 14 and dehydro-δ-viniferin 15) were endowed with significant antibacterial activity against gram-positive bacteria. The exposure of gram-positive foodborne pathogens to 100 µg/mL of 2, 9 and 15 induced severe cell membrane damage, resulting in the disruption of the phospholipid bilayer. The most promising dimeric compound, dehydro-δ-viniferin 15, was tested against Listeria monocytogenes, resulting in a loss of cultivability, viability and cell membrane potential. TEM analysis revealed grave morphological modifications on the cell membrane and leakage of intracellular content, confirming that the cell membrane was the principal biological target of the tested derivative.
Conflict of interest statement
The authors declare no competing interests.
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

