Site-Specific Hyperphosphorylation Inhibits, Rather than Promotes, Tau Fibrillization, Seeding Capacity, and Its Microtubule Binding
- PMID: 31863676
- PMCID: PMC7065254
- DOI: 10.1002/anie.201913001
Site-Specific Hyperphosphorylation Inhibits, Rather than Promotes, Tau Fibrillization, Seeding Capacity, and Its Microtubule Binding
Abstract
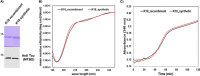
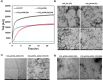
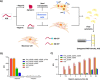
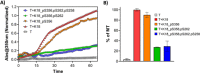
The consistent observation of phosphorylated tau in the pathology of Alzheimer's disease has contributed to the emergence of a model where hyperphosphorylation triggers both tau disassociation from microtubules and its subsequent aggregation. Herein, we applied a total chemical synthetic approach to site-specifically phosphorylate the microtubule binding repeat domain of tau (K18) at single (pS356) or multiple (pS356/pS262 and pS356/pS262/pS258) residues. We show that hyperphosphorylation of K18 inhibits 1) its aggregation in vitro, 2) its seeding activity in cells, 3) its binding to microtubules, and 4) its ability to promote microtubule polymerization. The inhibition increased with increasing the number of phosphorylated sites, with phosphorylation at S262 having the strongest effect. Our results argue against the hyperphosphorylation hypothesis and underscore the importance of revisiting the role of site-specific hyperphosphorylation in regulating tau functions in health and disease.
Keywords: aggregation; hyperphosphorylation; native state stabilization; protein modifications; tau protein.
© 2019 The Authors. Published by Wiley-VCH Verlag GmbH & Co. KGaA.
Conflict of interest statement
Hilal A. Lashuel is the founder and CSO of ND BioSciences.
Figures

Similar articles
-
Phosphorylation of the overlooked tyrosine 310 regulates the structure, aggregation, and microtubule- and lipid-binding properties of Tau.J Biol Chem. 2020 Jun 5;295(23):7905-7922. doi: 10.1074/jbc.RA119.012517. Epub 2020 Apr 27. J Biol Chem. 2020. PMID: 32341125 Free PMC article.
-
Phosphorylation that detaches tau protein from microtubules (Ser262, Ser214) also protects it against aggregation into Alzheimer paired helical filaments.Biochemistry. 1999 Mar 23;38(12):3549-58. doi: 10.1021/bi981874p. Biochemistry. 1999. PMID: 10090741
-
Structural and functional implications of tau hyperphosphorylation: information from phosphorylation-mimicking mutated tau proteins.Biochemistry. 2000 Oct 31;39(43):13166-75. doi: 10.1021/bi001290z. Biochemistry. 2000. PMID: 11052669
-
Tau domains, phosphorylation, and interactions with microtubules.Neurobiol Aging. 1995 May-Jun;16(3):355-62; discussion 362-3. doi: 10.1016/0197-4580(95)00025-a. Neurobiol Aging. 1995. PMID: 7566345 Review.
-
Natural products against tau hyperphosphorylation-induced aggregates: Potential therapies for Alzheimer's disease.Arch Pharm (Weinheim). 2025 Jan;358(1):e2400721. doi: 10.1002/ardp.202400721. Arch Pharm (Weinheim). 2025. PMID: 39888017 Free PMC article. Review.
Cited by
-
D-Peptide and D-Protein Technology: Recent Advances, Challenges, and Opportunities.Chembiochem. 2023 Feb 14;24(4):e202200537. doi: 10.1002/cbic.202200537. Epub 2022 Nov 16. Chembiochem. 2023. PMID: 36278392 Free PMC article. Review.
-
A Structural Ensemble of a Tau-Microtubule Complex Reveals Regulatory Tau Phosphorylation and Acetylation Mechanisms.ACS Cent Sci. 2021 Dec 22;7(12):1986-1995. doi: 10.1021/acscentsci.1c00585. Epub 2021 Dec 8. ACS Cent Sci. 2021. PMID: 34963892 Free PMC article.
-
Distinct tau folds initiate templated seeding and alter the post-translational modification profile.Brain. 2023 Dec 1;146(12):4988-4999. doi: 10.1093/brain/awad272. Brain. 2023. PMID: 37904205 Free PMC article.
-
Phosphorylation of the overlooked tyrosine 310 regulates the structure, aggregation, and microtubule- and lipid-binding properties of Tau.J Biol Chem. 2020 Jun 5;295(23):7905-7922. doi: 10.1074/jbc.RA119.012517. Epub 2020 Apr 27. J Biol Chem. 2020. PMID: 32341125 Free PMC article.
-
A Versatile Method for Site-Specific Chemical Installation of Aromatic Posttranslational Modification Analogs into Proteins.J Am Chem Soc. 2024 Sep 18;146(37):25788-25798. doi: 10.1021/jacs.4c08416. Epub 2024 Sep 3. J Am Chem Soc. 2024. PMID: 39224092 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

