Evaluating spatiotemporal microstructural alterations following diffuse traumatic brain injury
- PMID: 31865019
- PMCID: PMC6931220
- DOI: 10.1016/j.nicl.2019.102136
Evaluating spatiotemporal microstructural alterations following diffuse traumatic brain injury
Abstract
Background: Diffuse traumatic brain injury (TBI) is known to lead to microstructural changes within both white and grey matter detected in vivo with diffusion tensor imaging (DTI). Numerous studies have shown alterations in fractional anisotropy (FA) and mean diffusivity (MD) within prominent white matter tracts, but few have linked these to changes within the grey matter with confirmation via histological assessment. This is especially important as alterations in the grey matter may be predictive of long-term functional deficits.
Methods: A total of 33 male Sprague Dawley rats underwent severe closed-head TBI. Eight animals underwent tensor-based morphometry (TBM) and DTI at baseline (pre-TBI), 24 hours (24 h), 7, 14, and 30 days post-TBI. Immunohistochemical analysis for the detection of ionised calcium-binding adaptor molecule 1 (IBA1) to assess microglia number and percentage of activated cells, β-amyloid precursor protein (APP) as a marker of axonal injury, and myelin basic protein (MBP) to investigate myelination was performed at each time-point.
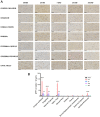
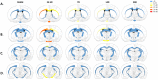
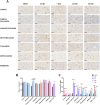
Results: DTI showed significant alterations in FA and RD in numerous white matter tracts including the corpus callosum, internal and external capsule, and optic tract and in the grey-matter in the cortex, thalamus, and hippocampus, with the most significant effects observed at 14 D post-TBI. TBM confirmed volumetric changes within the hippocampus and thalamus. Changes in DTI were in line with significant axonal injury noted at 24 h post-injury via immunohistochemical analysis of APP, with widespread microglial activation seen within prominent white matter tracts and the grey matter, which persisted to 30 D within the hippocampus and thalamus. Microstructural alterations in MBP+ve fibres were also noted within the hippocampus and thalamus, as well as the cortex.
Conclusion: This study confirms the widespread effects of diffuse TBI on white matter tracts which could be detected via DTI and extends these findings to key grey matter regions, with a comprehensive investigation of the whole brain. In particular, the hippocampus and thalamus appear to be vulnerable to ongoing pathology post-TBI, with DTI able to detect these alterations supporting the clinical utility in evaluating these regions post-TBI.
Keywords: Axonal injury; Diffusion tensor Imaging; Neuroinflammation; Structural magnetic resonance imaging; Traumatic brain injury.
Copyright © 2019. Published by Elsevier Inc.
Conflict of interest statement
Declaration of Competing Interest The authors report no competing interests.
Figures




References
-
- Ariza M., Serra-Grabulosa J.M., Junqué C., Ramírez B., Mataró M., Poca A., Bargalló N., Sahuquillo J. Hippocampal head atrophy after traumatic brain injury. Neuropsychologia. 2006;44:1956–1961. - PubMed
-
- Armstrong R.C., Mierzwa A.J., Marion C.M., Sullivan G.M. White matter involvement after TBI: clues to axon and myelin repair capacity. Exp. Neurol. 2016;275:328–333. - PubMed
-
- Arulsamy A., Teng J., Colton H., Corrigan F., Collins-Praino L. Evaluation of early chronic functional outcomes and their relationship to pre-frontal cortex and hippocampal pathology following moderate-severe traumatic brain injury. Behav. Brain Res. 2018;348:127–138. - PubMed
-
- Bareyre F., Wahl F., McIntosh T.K., Stutzmann J.M. Time course of cerebral edema after traumatic brain injury in rats: effects of riluzole and mannitol. J. Neurotrauma. 1997;14:839–849. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

