BST2 suppresses porcine epidemic diarrhea virus replication by targeting and degrading virus nucleocapsid protein with selective autophagy
- PMID: 31868081
- PMCID: PMC8386623
- DOI: 10.1080/15548627.2019.1707487
BST2 suppresses porcine epidemic diarrhea virus replication by targeting and degrading virus nucleocapsid protein with selective autophagy
Abstract
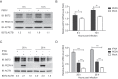
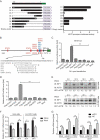
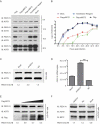
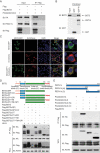
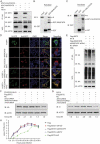
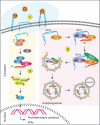
Interferon-induced BST2 (bone marrow stromal cell antigen 2) inhibits viral replication by tethering enveloped virions to the cell surface to restrict viral release and by inducing the NFKB-dependent antiviral immune response. However, the mechanism by which BST2 uses the selective autophagy pathway to inhibit viral replication is poorly understood. In this study, we showed that BST2 expression was significantly increased during porcine epidemic diarrhea virus (PEDV) infection of Vero cells by IRF1 targeting its promoter. We also showed that BST2 suppressed PEDV replication by binding and degrading the PEDV-encoded nucleocapsid (N) protein. The downregulation of N protein was blocked by macroautophagy/autophagy inhibitors but not a proteasome inhibitor, implying that the N protein was degraded via the selective autophagy pathway. Both the BST2 and N protein interacted with the E3 ubiquitin ligase MARCHF8/MARCH8 and the cargo receptor CALCOCO2/NDP52, and the ubiquitination of N protein was necessary for the degradation of N mediated by the BST2-MARCHF8 axis. The knockdown of MARCHF8 or ATG5 with small interfering RNAs blocked the selective autophagy pathway, rescued the protein abundance of PEDV N in 293T cells, and prevented the inhibition of PEDV replication by BST2 in Vero cells. Together, our data demonstrate the novel mechanism of BST2-mediated virus restriction, in which BST2 recruits MARCHF8 to catalyze the ubiquitination of the PEDV N protein. The ubiquitinated N protein is then recognized by CALCOCO2/NDP52, which delivers it to autolysosome for degradation through the selective autophagy pathway. Abbreviations: 3MA: 3-methyladenine; ATG: autophagy-related; Baf A1: bafilomycin A1; BST2: bone marrow stromal cell antigen 2; CALCOCO2/NDP52: calcium binding and coiled-coil domain 2; CC: coiled-coil; ChIP: chromatin immunoprecipitation; Co-IP: co-immunoprecipitation; CQ: chloroquine; CT: cytoplasmic tail; DAPI: 4',6-diamidino-2-phenylindole; GPI: glycosyl-phosphatidylinositol; hpi: hours post infection; IRF1: interferon regulatory factor 1; ISG: IFN-stimulated gene; MAP1LC3/LC3: microtubule associated protein 1 light chain 3; MARCHF8/MARCH8: membrane-associated ring-CH-type finger 8; MOI: multiplicity of infection; N protein: nucleocapsid protein; PED: porcine epidemic diarrhea; PEDV: porcine epidemic diarrhea virus; RT: room temperature; siRNA: small interfering RNA; STAT: signal transducer and activator of transcription; TCID50: 50% tissue culture infectious doses; TM: transmembrane.
Keywords: BST2; CALCOCO2/NDP52; IRF1; MARCHF8/MARCH8; PEDV; nucleocapsid protein; replication; selective autophagy.
Conflict of interest statement
No potential conflict of interest was reported by the authors.
Figures

Similar articles
-
N protein of PEDV plays chess game with host proteins by selective autophagy.Autophagy. 2023 Aug;19(8):2338-2352. doi: 10.1080/15548627.2023.2181615. Epub 2023 Mar 2. Autophagy. 2023. PMID: 36861818 Free PMC article.
-
FUBP3 Degrades the Porcine Epidemic Diarrhea Virus Nucleocapsid Protein and Induces the Production of Type I Interferon.J Virol. 2022 Jul 13;96(13):e0061822. doi: 10.1128/jvi.00618-22. Epub 2022 Jun 13. J Virol. 2022. PMID: 35695513 Free PMC article.
-
Enteric coronavirus nsp2 is a virulence determinant that recruits NBR1 for autophagic targeting of TBK1 to diminish the innate immune response.Autophagy. 2024 Aug;20(8):1762-1779. doi: 10.1080/15548627.2024.2340420. Epub 2024 Apr 16. Autophagy. 2024. PMID: 38597182 Free PMC article.
-
Coronavirus interactions with the cellular autophagy machinery.Autophagy. 2020 Dec;16(12):2131-2139. doi: 10.1080/15548627.2020.1817280. Epub 2020 Sep 23. Autophagy. 2020. PMID: 32964796 Free PMC article. Review.
-
Emerging views of OPTN (optineurin) function in the autophagic process associated with disease.Autophagy. 2022 Jan;18(1):73-85. doi: 10.1080/15548627.2021.1908722. Epub 2021 Apr 13. Autophagy. 2022. PMID: 33783320 Free PMC article. Review.
Cited by
-
A Comprehensive View on the Protein Functions of Porcine Epidemic Diarrhea Virus.Genes (Basel). 2024 Jan 26;15(2):165. doi: 10.3390/genes15020165. Genes (Basel). 2024. PMID: 38397155 Free PMC article. Review.
-
TRIM21 inhibits porcine epidemic diarrhea virus proliferation by proteasomal degradation of the nucleocapsid protein.Arch Virol. 2021 Jul;166(7):1903-1911. doi: 10.1007/s00705-021-05080-4. Epub 2021 Apr 26. Arch Virol. 2021. PMID: 33900472 Free PMC article.
-
Coxsackievirus B3 infection induces glycolysis to facilitate viral replication.Front Microbiol. 2022 Dec 9;13:962766. doi: 10.3389/fmicb.2022.962766. eCollection 2022. Front Microbiol. 2022. PMID: 36569097 Free PMC article.
-
Establishment and Verification of a Gene Signature for Diagnosing Type 2 Diabetics by WGCNA, LASSO Analysis, and In Vitro Experiments.Biomed Res Int. 2022 May 23;2022:4446342. doi: 10.1155/2022/4446342. eCollection 2022. Biomed Res Int. 2022. Retraction in: Biomed Res Int. 2024 Mar 20;2024:9828614. doi: 10.1155/2024/9828614. PMID: 35655479 Free PMC article. Retracted.
-
hnRNP K Degrades Viral Nucleocapsid Protein and Induces Type I IFN Production to Inhibit Porcine Epidemic Diarrhea Virus Replication.J Virol. 2022 Nov 23;96(22):e0155522. doi: 10.1128/jvi.01555-22. Epub 2022 Nov 1. J Virol. 2022. PMID: 36317879 Free PMC article.
References
-
- Debouck P.Pensaert M Experimental infection of pigs with a new porcine enteric coronavirus, CV 777. Am J Vet Res. 1980;41:219–223. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
