Erenumab safety and efficacy in migraine: A systematic review and meta-analysis of randomized clinical trials
- PMID: 31876735
- PMCID: PMC6946432
- DOI: 10.1097/MD.0000000000018483
Erenumab safety and efficacy in migraine: A systematic review and meta-analysis of randomized clinical trials
Abstract
Background: Erenumab is a new medicine recently approved in the United States of America for the preventive treatment of migraine among adults. We aimed to conduct a meta-analysis and evaluation of the efficacy and safety of erenumab among patients with migraine.
Methods: The electronic databases that were searched comprised PubMed, Embase and the Cochrane library, which were independently retrieved by 2 reviewers. Only randomized controlled trials (RCTs) that compared placebo with erenumab were selected. Mean differences (MDs), pooled risk ratios (RRs), and their corresponding 95% confidence intervals (CIs) were calculated for continuous and dichotomous data, respectively.
Results: Five RCTs representing 2928 patients were included. Pooled analysis showed significant reductions in the 50% responder rate (RR 1.55; P < .00001; I = 49%). In addition, the mean monthly migraine days from baseline in the erenumab group compared with placebo (MD-1.32; P < .00001; I = 100%) and migraine-specific medication days) from baseline (MD-1.41; P < .00001; I = 100%) were significantly decreased for the erenumab group as compared with placebo. Furthermore, Migraine-specific medication days from baseline in the 140 mg erenumab group were significantly reduced as compared the 70 mg group (MD = 0.55; P < .00001; I = 90%). Finally, there was no significant difference between the erenumab group and placebo for any adverse event and serious adverse event.
Conclusion: Among patients with migraine, both 70 and 140 mg of erenumab were associated with reduced Migraine-specific medication days, Migraine-specific medication days from baseline, and an increased rate of a 50% reduction, in the absence of an increased risk of any serious adverse effect.
Conflict of interest statement
This study and the analyses contained herein were supported by the National Key Specialty Construction Project of Clinical Pharmacy (Gran No. 30305030698). The funding was aligned to staff compensation accounting, budgeting the design of the study, and the collection, and interpretation of data and in drafting the manuscript. Authors declare that they have no real or perceived financial or academic conflicts of interest in regard the conduct of this work, or its synthesis and publication.
Figures



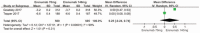

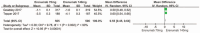

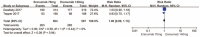

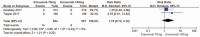
References
-
- Headache disorders. https://www.who.int/en/news-room/fact-sheets/detail/headache-disorders [accessed date October 14, 2019].
-
- Jain S, Yuan H, Spare N, et al. Erenumab in the treatment of migraine. Pain Manag 2018;8:415–26. - PubMed
-
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition. Cephalalgia 2018;38:1–211. - PubMed
-
- Lipton RB, Silberstein SD. Episodic and chronic migraine headache: breaking down barriers to optimal treatment and prevention. Headache 2015;55: Suppl 2: 103–22. quiz 123-6. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

