Nine years of in situ soil warming and topography impact the temperature sensitivity and basal respiration rate of the forest floor in a Canadian boreal forest
- PMID: 31877170
- PMCID: PMC6932772
- DOI: 10.1371/journal.pone.0226909
Nine years of in situ soil warming and topography impact the temperature sensitivity and basal respiration rate of the forest floor in a Canadian boreal forest
Abstract
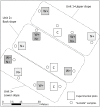
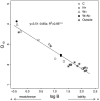
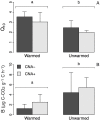
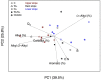
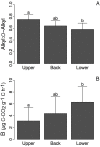
The forest floor of boreal forest stores large amounts of organic C that may react to a warming climate and increased N deposition. It is therefore crucial to assess the impact of these factors on the temperature sensitivity of this C pool to help predict future soil CO2 emissions from boreal forest soils to the atmosphere. In this study, soil warming (+2-4°C) and canopy N addition (CNA; +0.30-0.35 kg·N·ha-1·yr-1) were replicated along a topographic gradient (upper, back and lower slope) in a boreal forest in Quebec, Canada. After nine years of treatment, the forest floor was collected in each plot, and its organic C composition was characterized through solid-state 13C nuclear magnetic resonance (NMR) spectroscopy. Forest floor samples were incubated at four temperatures (16, 24, 32 and 40°C) and respiration rates (RR) measured to assess the temperature sensitivity of forest floor RR (Q10 = e10k) and basal RR (B). Both soil warming and CNA had no significant effect on forest floor chemistry (e.g., C, N, Ca and Mg content, amount of soil organic matter, pH, chemical functional groups). The NMR analyses did not show evidence of significant changes in the forest floor organic C quality. Nonetheless, a significant effect of soil warming on both the Q10 of RR and B was observed. On average, B was 72% lower and Q10 45% higher in the warmed, versus the control plots. This result implies that forest floor respiration will more strongly react to changes in soil temperature in a future warmer climate. CNA had no significant effect on the measured soil and respiration parameters, and no interaction effects with warming. In contrast, slope position had a significant effect on forest floor organic C quality. Upper slope plots had higher soil alkyl C:O-alkyl C ratios and lower B values than those in the lower slope, across all different treatments. This result likely resulted from a relative decrease in the labile C fraction in the upper slope, characterized by lower moisture levels. Our results point towards higher temperature sensitivity of RR under warmer conditions, accompanied by an overall down-regulation of RR at low temperatures (lower B). Since soil C quantity and quality were unaffected by the nine years of warming, the observed patterns could result from microbial adaptations to warming.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

Similar articles
-
Microbial physiology and soil CO2 efflux after 9 years of soil warming in a temperate forest - no indications for thermal adaptations.Glob Chang Biol. 2015 Nov;21(11):4265-77. doi: 10.1111/gcb.12996. Epub 2015 Sep 28. Glob Chang Biol. 2015. PMID: 26046333 Free PMC article.
-
Carbon quality and soil microbial property control the latitudinal pattern in temperature sensitivity of soil microbial respiration across Chinese forest ecosystems.Glob Chang Biol. 2018 Jul;24(7):2841-2849. doi: 10.1111/gcb.14105. Epub 2018 Mar 25. Glob Chang Biol. 2018. PMID: 29476638
-
Interactive biotic and abiotic regulators of soil carbon cycling: evidence from controlled climate experiments on peatland and boreal soils.Glob Chang Biol. 2014 Sep;20(9):2971-82. doi: 10.1111/gcb.12585. Epub 2014 Apr 26. Glob Chang Biol. 2014. PMID: 24687903
-
How do forest fires affect soil greenhouse gas emissions in upland boreal forests? A review.Environ Res. 2020 May;184:109328. doi: 10.1016/j.envres.2020.109328. Epub 2020 Mar 5. Environ Res. 2020. PMID: 32163772 Review.
-
Influences of evergreen gymnosperm and deciduous angiosperm tree species on the functioning of temperate and boreal forests.Biol Rev Camb Philos Soc. 2015 May;90(2):444-66. doi: 10.1111/brv.12119. Epub 2014 Jun 11. Biol Rev Camb Philos Soc. 2015. PMID: 24916992 Review.
References
-
- Davidson EA, Janssens IA. Temperature sensitivity of soil carbon decomposition and feedbacks to climate change. Nature [Internet]. 2006. March 9 [cited 2011 Jun 11];440(7081):165–73. Available from: http://www.ncbi.nlm.nih.gov/pubmed/16525463 10.1038/nature04514 - DOI - PubMed
-
- Conant RT, Ryan MG, Ågren GI, Birge HE, Davidson EA., Eliasson PE, et al. Temperature and soil organic matter decomposition rates—synthesis of current knowledge and a way forward. Glob Chang Biol [Internet]. 2011. Nov 2 [cited 2013 Oct 22];17(11):3392–404. Available from: http://doi.wiley.com/10.1111/j.1365-2486.2011.02496.x - DOI
-
- Kirschbaum MUFF. The temperature dependence of soil organic matter decomposition, and the effect of global warming on soil organic C storage. Soil Biol Biochem [Internet]. 1995;27(6):753–60. Available from: http://linkinghub.elsevier.com/retrieve/pii/003807179400242S
-
- Melillo JM, Frey SD, Deangelis KM, Werner WJ, Bernard MJ, Bowles FP, et al. Long-term pattern and magnitude of soil carbon feedback to the climate system in a warming world. Science (80-). 2017;358:101–5. - PubMed
-
- Hamdi S, Moyano F, Sall S, Bernoux M, Chevallier T. Synthesis analysis of the temperature sensitivity of soil respiration from laboratory studies in relation to incubation methods and soil conditions. Soil Biol Biochem [Internet]. 2013;58:115–26. Available from: 10.1016/j.soilbio.2012.11.012 - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

