Antirotavirus IgA seroconversion rates in children who receive concomitant oral poliovirus vaccine: A secondary, pooled analysis of Phase II and III trial data from 33 countries
- PMID: 31887139
- PMCID: PMC6936798
- DOI: 10.1371/journal.pmed.1003005
Antirotavirus IgA seroconversion rates in children who receive concomitant oral poliovirus vaccine: A secondary, pooled analysis of Phase II and III trial data from 33 countries
Abstract
Background: Despite the success of rotavirus vaccines over the last decade, rotavirus remains a leading cause of severe diarrheal disease among young children. Further progress in reducing the burden of disease is inhibited, in part, by vaccine underperformance in certain settings. Early trials suggested that oral poliovirus vaccine (OPV), when administered concomitantly with rotavirus vaccine, reduces rotavirus seroconversion rates after the first rotavirus dose with modest or nonsignificant interference after completion of the full rotavirus vaccine course. Our study aimed to identify a range of individual-level characteristics, including concomitant receipt of OPV, that affect rotavirus vaccine immunogenicity in high- and low-child-mortality settings, controlling for individual- and country-level factors. Our central hypothesis was that OPV administered concomitantly with rotavirus vaccine reduced rotavirus vaccine immunogenicity.
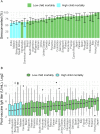
Methods and findings: Pooled, individual-level data from GlaxoSmithKline's Phase II and III clinical trials of the monovalent rotavirus vaccine (RV1), Rotarix, were analyzed, including 7,280 vaccinated infants (5-17 weeks of age at first vaccine dose) from 22 trials and 33 countries/territories (5 countries/territories with high, 13 with moderately low, and 15 with very low child mortality). Two standard markers for immune response were examined including antirotavirus immunoglobulin A (IgA) seroconversion (defined as the appearance of serum antirotavirus IgA antibodies in subjects initially seronegative) and serum antirotavirus IgA titer, both collected approximately 4-12 weeks after administration of the last rotavirus vaccine dose. Mixed-effect logistic regression and mixed-effect linear regression of log-transformed data were used to identify individual- and country-level predictors of seroconversion (dichotomous) and antibody titer (continuous), respectively. Infants in high-child-mortality settings had lower odds of seroconverting compared with infants in low-child-mortality settings (odds ratio [OR] = 0.48, 95% confidence interval [CI] 0.43-0.53, p < 0.001). Similarly, among those who seroconverted, infants in high-child-mortality settings had lower IgA titers compared with infants in low-child-mortality settings (mean difference [β] = 0.83, 95% CI 0.77-0.90, p < 0.001). Infants who received OPV concomitantly with both their first and their second doses of rotavirus vaccine had 0.63 times the odds of seroconverting (OR = 0.63, 95% CI 0.47-0.84, p = 0.002) compared with infants who received OPV but not concomitantly with either dose. In contrast, among infants who seroconverted, OPV concomitantly administered with both the first and second rotavirus vaccine doses was found to be positively associated with antirotavirus IgA titer (β = 1.28, 95% CI 1.07-1.53, p = 0.009). Our findings may have some limitations in terms of generalizability to routine use of rotavirus vaccine because the analysis was limited to healthy infants receiving RV1 in clinical trial settings.
Conclusions: Our findings suggest that OPV given concomitantly with RV1 was a substantial contributor to reduced antirotavirus IgA seroconversion, and this interference was apparent after the second vaccine dose of RV1, as with the original clinical trials that our reanalysis is based on. However, our findings do suggest that the forthcoming withdrawal of OPV from the infant immunization schedule globally has the potential to improve RV1 performance.
Conflict of interest statement
I have read the journal's policy and the authors of this manuscript have the following competing interests: BAL has received personal fees from Takeda Pharmaceuticals, personal fees from CDC Foundation, and personal fees from Hall Booth Smith, P.C., outside the submitted work. All other authors declare no competing interests.
Figures
References
-
- Centers for Disease Control and Prevention [Internet]. Atlanta, GA: Centers for Diesease Control and Prevention; [cited 2017 Aug 20]. Rotavirus and the Vaccine (Drops) to Prevent It. Available from: https://www.cdc.gov/vaccines/parents/diseases/child/rotavirus.html.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

