Preclinical evaluation of [18F]FB-A20FMDV2 as a selective marker for measuring αVβ6 integrin occupancy using positron emission tomography in rodent lung
- PMID: 31897589
- PMCID: PMC7075836
- DOI: 10.1007/s00259-019-04653-5
Preclinical evaluation of [18F]FB-A20FMDV2 as a selective marker for measuring αVβ6 integrin occupancy using positron emission tomography in rodent lung
Abstract
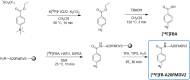
Purpose: Integrin αvβ6 belongs to the RGD subset of the integrin family, and its expression levels are a prognostic and theranostic factor in some types of cancer and pulmonary fibrosis. This paper describes the GMP radiolabelling of the synthetic 20 amino acid peptide A20FMDV2 (NAVPNLRGDLQVLAQKVART), derived from the foot-and-mouth disease virus, and characterises the use of [18F]FB-A20FMDV2 as a high affinity, specific and selective PET radioligand for the quantitation and visualisation of αvβ6 in rodent lung to support human translational studies.
Methods: The synthesis of [18F]FB-A20FMDV2 was performed using a fully automated and GMP-compliant process. Sprague-Dawley rats were used to perform homologous (unlabelled FB-A20FMDV2) and heterologous (anti-αvβ6 antibody 8G6) blocking studies. In order to generate a dosimetry estimate, tissue residence times were generated, and associated tissue exposure and effective dose were calculated using the Organ Level Internal Dose Assessment/Exponential Modelling (OLINDA/EXM) software.
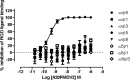
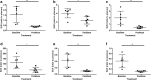
Results: [18F]FB-A20FMDV2 synthesis was accomplished in 180 min providing ~800 MBq of [18F]FB-A20FMDV2 with a molar activity of up to 150 GBq/μmol and high radiochemical purity (> 97%). Following i.v. administration to rats, [18F]FB-A20FMDV2 was rapidly metabolised with intact radiotracer representing 5% of the total radioactivity present in rat plasma at 30 min. For the homologous and heterologous block in rats, lung-to-heart SUV ratios at 30-60 min post-administration of [18F]FB-A20FMDV2 were reduced by 38.9 ± 6.9% and 56 ± 19.2% for homologous and heterologous block, respectively. Rodent biodistribution and dosimetry calculations using OLINDA/EXM provided a whole body effective dose in humans 33.5 μSv/MBq.
Conclusion: [18F]FB-A20FMDV2 represents a specific and selective PET ligand to measure drug-associated αvβ6 integrin occupancy in lung. The effective dose, extrapolated from rodent data, is in line with typical values for compounds labelled with fluorine-18 and combined with the novel fully automated and GMP-compliant synthesis and allows for clinical use in translational studies.
Keywords: A20FMDV2; Fibrosis; Integrin; PET; Rodent lung; αvβ6.
Figures



References
-
- Breuss J, Gallo J, DeLisser H, Klimanskaya I, Folkesson H, Pittet J, et al. Expression of the beta 6 integrin subunit in development, neoplasia and tissue repair suggests a role in epithelial remodeling. J Cell Sci. 1995;108(6):2241–2251. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

