The 111 Study: A Single-arm, Phase 3 Trial Evaluating One Cycle of Bleomycin, Etoposide, and Cisplatin as Adjuvant Chemotherapy in High-risk, Stage 1 Nonseminomatous or Combined Germ Cell Tumours of the Testis
- PMID: 31901440
- PMCID: PMC7026695
- DOI: 10.1016/j.eururo.2019.11.022
The 111 Study: A Single-arm, Phase 3 Trial Evaluating One Cycle of Bleomycin, Etoposide, and Cisplatin as Adjuvant Chemotherapy in High-risk, Stage 1 Nonseminomatous or Combined Germ Cell Tumours of the Testis
Abstract
Background: Standard management in the UK for high-risk stage 1 nonseminoma germ cell tumours of the testis (NSGCTT) is two cycles of adjuvant bleomycin, etoposide (360 mg/m2), and cisplatin (BE360P) chemotherapy, or surveillance.
Objective: To test whether one cycle of BE500P achieves similar recurrence rates to two cycles of BE360P.
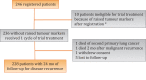
Design, setting, and participants: A total of 246 patients with vascular invasion-positive stage 1 NSGCTT or combined seminoma + NSGCTT were centrally registered in a single-arm prospective study.
Intervention: One cycle comprising bleomycin 30000 IU on days 1, 8, and 15, etoposide 165 mg/m2 on days 1-3, and cisplatin 50 mg/m2 on days 1-2, plus antibacterial and granulocyte colony stimulating factor prophylaxis.
Outcome measurements and statistical analysis: The primary endpoint was 2-yr malignant recurrence (MR); the aim was to exclude a rate of ≥5%. Participants had regular imaging and tumour marker (TM) assessment for 5 yr.
Results and limitations: The median follow-up was 49 mo (interquartile range 37-60). Ten patients with rising TMs at baseline were excluded. Four patients had MR at 6, 7, 13, and 27 mo; all received second-line chemotherapy and surgery and three remained recurrence-free at 5 yr. The 2-yr MR rate was 1.3% (95% confidence interval 0.3-3.7%). Three patients developed nonmalignant recurrences with localised teratoma differentiated, rendered disease-free after surgery. Grade 3-4 febrile neutropenia occurred in 6.8% of participants.
Conclusions: BE500P is safe and the 2-yr MR rate is consistent with that seen following two BE360P cycles. The 111 study is the largest prospective trial investigating one cycle of adjuvant BE500P in high-risk stage 1 NSGCTT. Adoption of one cycle of BE500P as standard would reduce overall exposure to chemotherapy in this young population.
Patient summary: Removing the testicle fails to cure many patients with high-risk primary testicular cancer since undetectable cancers are often present elsewhere. A standard additional treatment in Europe is two cycles of chemotherapy to eradicate these. This trial shows one cycle has few adverse effects and comparable outcomes to those seen with two cycles.
Keywords: Adjuvant therapy; BEP; Chemotherapy; NSGCTT; Phase III trial; Surveillance; Testicular cancer.
Copyright © 2019 The Authors. Published by Elsevier B.V. All rights reserved.
Figures
Comment in
-
The New Standard Adjuvant Treatment for High-risk Stage 1 Nonseminoma: Reducing the Treatment Burden and Maximizing Long-term Survival.Eur Urol. 2020 Mar;77(3):352-353. doi: 10.1016/j.eururo.2019.12.010. Epub 2019 Dec 20. Eur Urol. 2020. PMID: 31870512 No abstract available.
-
Zero, Some Chemotherapy Game in Early-Stage Germ Cell Tumors.J Clin Oncol. 2020 Jul 1;38(19):2214-2215. doi: 10.1200/JCO.20.00204. Epub 2020 Apr 22. J Clin Oncol. 2020. PMID: 32320312 No abstract available.
-
Reply to Torgrim Tandstad. The New Standard Adjuvant Treatment for High-risk Stage 1 Nonseminoma: Reducing the Treatment Burden and Maximizing Long-term Survival. Eur Urol 2020;77:352-3.Eur Urol. 2020 Dec;78(6):e220. doi: 10.1016/j.eururo.2020.05.023. Epub 2020 Jun 7. Eur Urol. 2020. PMID: 32522389 No abstract available.
-
Re: The 111 Study: A Single-arm, Phase 3 Trial Evaluating One Cycle of Bleomycin, Etoposide, and Cisplatin as Adjuvant Chemotherapy in High-risk, Stage 1 Nonseminomatous or Combined Germ Cell Tumours of the Testis.Eur Urol. 2021 Jan;79(1):160-161. doi: 10.1016/j.eururo.2020.08.010. Epub 2020 Nov 20. Eur Urol. 2021. PMID: 33229103 No abstract available.
Similar articles
-
Long-term outcome of patients with clinical stage I high-risk nonseminomatous germ-cell tumors 15 years after one adjuvant cycle of bleomycin, etoposide, and cisplatin chemotherapy.Ann Oncol. 2015 Feb;26(2):374-7. doi: 10.1093/annonc/mdu518. Epub 2014 Nov 12. Ann Oncol. 2015. PMID: 25392157 Clinical Trial.
-
Surveillance or adjuvant treatments in stage 1 testis germ-cell tumours.Ann Oncol. 2012 Sep;23 Suppl 10:x342-8. doi: 10.1093/annonc/mds306. Ann Oncol. 2012. PMID: 22987989
-
Long-term followup results of 1 cycle of adjuvant bleomycin, etoposide and cisplatin chemotherapy for high risk clinical stage I nonseminomatous germ cell tumors of the testis.J Urol. 2008 Jan;179(1):163-6. doi: 10.1016/j.juro.2007.08.172. Epub 2007 Nov 14. J Urol. 2008. PMID: 18001800 Clinical Trial.
-
Long-term efficacy of two cycles of BEP regimen in high-risk stage I nonseminomatous testicular germ cell tumors with embryonal carcinoma and/or vascular invasion.Eur Urol. 2004 Aug;46(2):209-14; discussion 214-5. doi: 10.1016/j.eururo.2004.03.022. Eur Urol. 2004. PMID: 15245815 Review.
-
Long-term results following adjuvant chemotherapy in patients with clinical stage I testicular nonseminomatous malignant germ cell tumors with high risk factors.J Urol. 1999 Apr;161(4):1148-52. J Urol. 1999. PMID: 10081858 Review.
Cited by
-
Role of Epigenetics for the Efficacy of Cisplatin.Int J Mol Sci. 2024 Jan 17;25(2):1130. doi: 10.3390/ijms25021130. Int J Mol Sci. 2024. PMID: 38256203 Free PMC article. Review.
-
Canadian Urological Association consensus guideline: Management of testicular germ cell cancer.Can Urol Assoc J. 2022 Jun;16(6):155-173. doi: 10.5489/cuaj.7945. Can Urol Assoc J. 2022. PMID: 35623007 Free PMC article. No abstract available.
-
New insights on testicular cancer prevalence with novel diagnostic biomarkers and therapeutic approaches.Cancer Rep (Hoboken). 2024 Mar;7(3):e2052. doi: 10.1002/cnr2.2052. Cancer Rep (Hoboken). 2024. PMID: 38507271 Free PMC article. Review.
-
Editorial by Bendu K. Konneh, John T. Lafin and Aditya Bagrodia on pp. 341-342 of this issue: MicroRNA-371a-3p as a blood-based biomarker in testis cancer.Asian J Urol. 2021 Oct;8(4):400-406. doi: 10.1016/j.ajur.2021.08.004. Epub 2021 Aug 26. Asian J Urol. 2021. PMID: 34765447 Free PMC article. Review.
-
Subinguinal Orchidectomy for Testicular Cancer: Innovation or Unnecessary Advancement.Cureus. 2025 Jun 15;17(6):e86062. doi: 10.7759/cureus.86062. eCollection 2025 Jun. Cureus. 2025. PMID: 40666550 Free PMC article. Review.
References
-
- Freedman L.S., Parkinson M.C., Jones W.G. Histopathology in the prediction of relapse of patients with stage I testicular teratoma treated by orchidectomy alone. Lancet. 1987;2:294–298. - PubMed
-
- Read G., Stenning S.P., Cullen M.H. Medical Research Council prospective study of surveillance for stage I testicular teratoma. Medical Research Council Testicular Tumors Working Party. J Clin Oncol. 1992;10:1762–1768. - PubMed
-
- Vergouwe Y., Steyerberg E.W., Eijkemans M.J., Albers P., Habbema J.D. Predictors of occult metastasis in clinical stage I nonseminoma: a systematic review. J Clin Oncol. 2003;21:4092–4099. - PubMed
-
- Cullen M.H., Stenning S.P., Parkinson M.C. Short-course adjuvant chemotherapy in high-risk stage I nonseminomatous germ cell tumors of the testis: a Medical Research Council report. J Clin Oncol. 1996;14:1106–1113. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical