A new derivative of acetylsalicylic acid and carnosine: synthesis, physical and chemical properties, biological activity
- PMID: 31902097
- PMCID: PMC7214588
- DOI: 10.1007/s40199-019-00323-x
A new derivative of acetylsalicylic acid and carnosine: synthesis, physical and chemical properties, biological activity
Abstract
Purpose: The aim of this study was to create and assess biological activity of a new compound based on carnosine and acetylsalicylic acid (ASA) that will comprise antioxidant effect with antiplatelet activity, while simultaneously preventing side effects on the gastrointestinal tract.
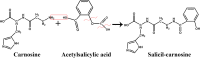
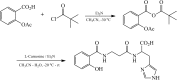
Methods: Salicyl-carnosine (SC) was synthesized by condensation of ASA and carnosine. Antioxidant activity was determined by spectrophotometric and chemiluminescence methods. Antiplatelet activity was carried out by the light transmission-aggregometry method using the inductor ADP. Chronic gastric ulcer in rats was modeled using glacial acetic acid.
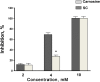
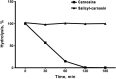
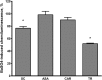
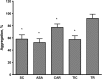
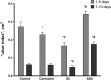
Results: Using SOD-like activity, iron-induced chemiluminescence, BaSO4-activated respiratory burst, and evaluation of red blood cell structure stabilization during oxidative damage induced by sodium hypochlorite, it was shown that SC possesses antioxidant activity analogous, or better, than that of carnosine. Antiplatelet activity of SC was evaluated in the blood of healthy individuals, and was also shown to be comparable to, or exceeding that of ASA. Also SC demonstrates high resistance to hydrolysis by tissue and serum carnosinases. Most importantly, it was shown that SC has protected the gastric mucosa against the formation of stomach ulcerative lesions and promoted their epithelization, therefore overcoming the undesirable inherent side effects of ASA.
Conclusions: SC preserves pharmacologically significant properties of ASA and carnosine while retaining an anti-ulcer activity and resistance to the carnosinase hydrolysis at the same time. These properties are particularly promising for the potential development of new anti-inflammatory and antithrombotic drugs. Graphical abstract .
Keywords: Acetylsalicylic acid; Antioxidant; Antiplatelet action; Carnosine; Gastrointestinal ulcer; Salicylic acid.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
An overview of the pathogenic mechanisms involved in severe cases of COVID-19 infection, and the proposal of salicyl-carnosine as a potential drug for its treatment.Eur J Pharmacol. 2020 Nov 5;886:173457. doi: 10.1016/j.ejphar.2020.173457. Epub 2020 Aug 1. Eur J Pharmacol. 2020. PMID: 32750366 Free PMC article. Review.
-
Synthesis and antiplatelet effects of the new antithrombotic agent aspalatone with low ulcerogenicity.Arzneimittelforschung. 1994 Oct;44(10):1122-6. Arzneimittelforschung. 1994. PMID: 7818584
-
Effects of carnosine and related compounds on the stability and morphology of erythrocytes from alcoholics.Alcohol Alcohol. 2000 Jan;35(1):44-8. doi: 10.1093/alcalc/35.1.44. Alcohol Alcohol. 2000. PMID: 10684775
-
Residence time of polaprezinc (zinc L-carnosine complex) in the rat stomach and adhesiveness to ulcerous sites.Jpn J Pharmacol. 1995 Apr;67(4):271-8. doi: 10.1254/jjp.67.271. Jpn J Pharmacol. 1995. PMID: 7650862
-
Enteric coating can lead to reduced antiplatelet effect of low-dose acetylsalicylic acid.Basic Clin Pharmacol Toxicol. 2015 Mar;116(3):212-5. doi: 10.1111/bcpt.12362. Epub 2014 Dec 23. Basic Clin Pharmacol Toxicol. 2015. PMID: 25469781 Review.
Cited by
-
Carnosine to Combat Novel Coronavirus (nCoV): Molecular Docking and Modeling to Cocrystallized Host Angiotensin-Converting Enzyme 2 (ACE2) and Viral Spike Protein.Molecules. 2020 Nov 28;25(23):5605. doi: 10.3390/molecules25235605. Molecules. 2020. PMID: 33260592 Free PMC article.
-
An overview of the pathogenic mechanisms involved in severe cases of COVID-19 infection, and the proposal of salicyl-carnosine as a potential drug for its treatment.Eur J Pharmacol. 2020 Nov 5;886:173457. doi: 10.1016/j.ejphar.2020.173457. Epub 2020 Aug 1. Eur J Pharmacol. 2020. PMID: 32750366 Free PMC article. Review.
-
Synthesis of Deuterium-Labeled Pyrrolylcarnosine.Dokl Biochem Biophys. 2022 Dec;507(1):374-379. doi: 10.1134/S1607672922340130. Epub 2023 Feb 14. Dokl Biochem Biophys. 2022. PMID: 36787006
-
Malondialdehyde as an Important Key Factor of Molecular Mechanisms of Vascular Wall Damage under Heart Diseases Development.Int J Mol Sci. 2022 Dec 21;24(1):128. doi: 10.3390/ijms24010128. Int J Mol Sci. 2022. PMID: 36613568 Free PMC article. Review.
-
Small-molecule metabolome identifies potential therapeutic targets against COVID-19.Sci Rep. 2022 Jun 15;12(1):10029. doi: 10.1038/s41598-022-14050-y. Sci Rep. 2022. PMID: 35705626 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

