Live births after oocyte in vitro maturation with a prematuration step in women with polycystic ovary syndrome
- PMID: 31902102
- PMCID: PMC7056678
- DOI: 10.1007/s10815-019-01677-6
Live births after oocyte in vitro maturation with a prematuration step in women with polycystic ovary syndrome
Abstract
Purpose: Standard oocyte in vitro maturation (IVM) usually results in lower pregnancy rates than in vitro fertilization (IVF). IVM preceded by a prematuration step improves the acquisition of oocyte developmental competence and can enhance embryo quality (EQ). This study evaluated the effectiveness of a biphasic culture system incorporating prematuration and IVM steps (CAPA-IVM) versus standard IVM in women with polycystic ovarian morphology (PCOM).
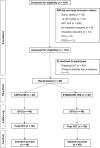
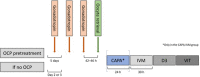
Methods: Eighty women (age < 38 years, ≥ 25 follicles of 2-9 mm in both ovaries, no major uterine abnormalities) were randomized to undergo CAPA-IVM (n = 40) or standard IVM (n = 40). CAPA-IVM uses two steps: a 24-h prematuration step with C-type natriuretic peptide-supplemented medium, then 30 h of culture in IVM media supplemented with follicle-stimulating hormone and amphiregulin. Standard IVM was performed using routine protocols.
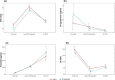
Results: A significantly higher proportion of oocytes reached metaphase II at 30 h after CAPA-IVM versus standard IVM (63.6 vs 49.0; p < 0.001) and the number of good quality embryos per cumulus-oocyte complex tended to be higher (18.9 vs 12.7; p = 0.11). Clinical pregnancy rate per embryo transfer was 63.2% in the CAPA-IVM versus 38.5% in the standard IVM group (p = 0.04). Live birth rate per embryo transfer was not statistically different between the CAPA-IVM and standard IVM groups (50.0 vs 33.3% [p = 0.17]). No malformations were reported and birth weight was similar in the two treatment groups.
Conclusions: Use of the CAPA-IVM system significantly improved maturation and clinical pregnancy rates versus standard IVM in patients with PCOM. Furthermore, live births after CAPA-IVM are reported for the first time.
Keywords: C-type natriuretic peptide; In vitro fertilization; In vitro maturation; Oocyte prematuration; Polycystic ovary syndrome.
Conflict of interest statement
All authors have no conflicts of interest to declare.
Figures

References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

