Quantitative Proteomics Reveal an Altered Pattern of Protein Expression in Brain Tissue from Mice Lacking GPR37 and GPR37L1
- PMID: 31903766
- PMCID: PMC7063848
- DOI: 10.1021/acs.jproteome.9b00622
Quantitative Proteomics Reveal an Altered Pattern of Protein Expression in Brain Tissue from Mice Lacking GPR37 and GPR37L1
Abstract
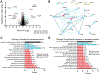
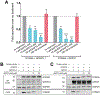
GPR37 and GPR37L1 are glia-enriched G protein-coupled receptors that have been implicated in several neurological and neurodegenerative diseases. To gain insight into the potential molecular mechanisms by which GPR37 and GPR37L1 regulate cellular physiology, proteomic analyses of whole mouse brain tissue from wild-type (WT) versus GPR37/GPR37L1 double knockout (DKO) mice were performed in order to identify proteins regulated by the absence versus presence of these receptors (data are available via ProteomeXchange with identifier PXD015202). These analyses revealed a number of proteins that were significantly increased or decreased by the absence of GPR37 and GPR37L1. One of the most decreased proteins in the DKO versus WT brain tissue was S100A5, a calcium-binding protein, and the reduction of S100A5 expression in KO brain tissue was validated via Western blot. Coexpression of S100A5 with either GPR37 or GPR37L1 in HEK293T cells did not result in any change in S100A5 expression but did robustly increase secretion of S100A5. To dissect the mechanism by which S100A5 secretion was enhanced, cells coexpressing S100A5 with the receptors were treated with different pharmacological reagents. These studies revealed that calcium is essential for the secretion of S100A5 downstream of GPR37 and GPR37L1 signaling, as treatment with BAPTA-AM, an intracellular Ca2+ chelator, reduced S100A5 secretion from transfected HEK293T cells. Collectively, these findings provide a panoramic view of proteomic changes resulting from loss of GPR37 and GPR37L1 and also impart mechanistic insight into the regulation of S100A5 by these receptors, thereby shedding light on the functions of GPR37 and GPR37L1 in brain tissue.
Keywords: S100A5; astrocyte; calcium; calcium-binding protein; glia; oligodendrocyte; secretion.
Figures



Similar articles
-
Mouse Mutants of Gpr37 and Gpr37l1 Receptor Genes: Disease Modeling Applications.Int J Mol Sci. 2022 Apr 13;23(8):4288. doi: 10.3390/ijms23084288. Int J Mol Sci. 2022. PMID: 35457105 Free PMC article. Review.
-
Mice lacking Gpr37 exhibit decreased expression of the myelin-associated glycoprotein MAG and increased susceptibility to demyelination.Neuroscience. 2017 Sep 1;358:49-57. doi: 10.1016/j.neuroscience.2017.06.006. Epub 2017 Jun 20. Neuroscience. 2017. PMID: 28642167 Free PMC article.
-
GPR37 and GPR37L1 are receptors for the neuroprotective and glioprotective factors prosaptide and prosaposin.Proc Natl Acad Sci U S A. 2013 Jun 4;110(23):9529-34. doi: 10.1073/pnas.1219004110. Epub 2013 May 20. Proc Natl Acad Sci U S A. 2013. PMID: 23690594 Free PMC article.
-
Glio- and neuro-protection by prosaposin is mediated by orphan G-protein coupled receptors GPR37L1 and GPR37.Glia. 2018 Nov;66(11):2414-2426. doi: 10.1002/glia.23480. Epub 2018 Sep 27. Glia. 2018. PMID: 30260505 Free PMC article.
-
Drug Discovery Opportunities at the Endothelin B Receptor-Related Orphan G Protein-Coupled Receptors, GPR37 and GPR37L1.Front Pharmacol. 2015 Nov 17;6:275. doi: 10.3389/fphar.2015.00275. eCollection 2015. Front Pharmacol. 2015. PMID: 26635605 Free PMC article. Review.
Cited by
-
Mouse Mutants of Gpr37 and Gpr37l1 Receptor Genes: Disease Modeling Applications.Int J Mol Sci. 2022 Apr 13;23(8):4288. doi: 10.3390/ijms23084288. Int J Mol Sci. 2022. PMID: 35457105 Free PMC article. Review.
-
Orphan G Protein-Coupled Receptor GPR37 as an Emerging Therapeutic Target.ACS Chem Neurosci. 2023 Sep 20;14(18):3318-3334. doi: 10.1021/acschemneuro.3c00479. Epub 2023 Sep 7. ACS Chem Neurosci. 2023. PMID: 37676000 Free PMC article. Review.
-
Combined Inhibition of AKT and KIT Restores Expression of Programmed Cell Death 4 (PDCD4) in Gastrointestinal Stromal Tumor.Cancers (Basel). 2021 Jul 23;13(15):3699. doi: 10.3390/cancers13153699. Cancers (Basel). 2021. PMID: 34359600 Free PMC article.
-
Impaired Aversive Memory Formation in GPR37L1KO Mice.Int J Mol Sci. 2022 Nov 18;23(22):14290. doi: 10.3390/ijms232214290. Int J Mol Sci. 2022. PMID: 36430766 Free PMC article.
-
The Effect of Early Life Stress on Emotional Behaviors in GPR37KO Mice.Int J Mol Sci. 2021 Dec 30;23(1):410. doi: 10.3390/ijms23010410. Int J Mol Sci. 2021. PMID: 35008836 Free PMC article.
References
-
- Cahoy JD; Emery B; Kaushal A; Foo LC; Zamanian JL; Christopherson KS; Xing Y; Lubischer JL; Krieg PA; Krupenko SA; Thompson WJ; Barres BA A transcriptome database for astrocytes, neurons, and oligodendrocytes: A new resource for understanding brain development and function. J. Neurosci 2008, 28, 264–78. - PMC - PubMed
-
- Marazziti D; Golini E; Gallo A; Lombardi MS; Matteoni R; Tocchini-Valentini GP Cloning of GPR37, a gene located on chromosome 7 encoding a putative G-protein-coupled receptor, from a human frontal brain EST library. Genomics, 1997, 45, 68–77. - PubMed
-
- Marazziti D; Gallo A, Golini E; Matteoni R; Tocchini-Valentini GP Molecular cloning and chromosomal localization of the mouse Gpr37 gene encoding an orphan G protein-coupled peptide receptor expressed in brain and testis. Genomics, 1998, 53, 315–24. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

