Cancer Associated Fibroblasts and Senescent Thyroid Cells in the Invasive Front of Thyroid Carcinoma
- PMID: 31906302
- PMCID: PMC7016563
- DOI: 10.3390/cancers12010112
Cancer Associated Fibroblasts and Senescent Thyroid Cells in the Invasive Front of Thyroid Carcinoma
Abstract
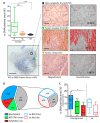
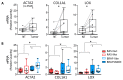
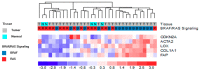
Thyroid carcinoma (TC) comprises several histotypes with different aggressiveness, from well (papillary carcinoma, PTC) to less differentiated forms (poorly differentiated and anaplastic thyroid carcinoma, PDTC and ATC, respectively). Previous reports have suggested a functional role for cancer-associated fibroblasts (CAFs) or senescent TC cells in the progression of PTC. In this study, we investigated the presence of CAFs and senescent cells in proprietary human TCs including PTC, PDTC, and ATC. Screening for the driving lesions BRAFV600E and N/H/KRAS mutations, and gene fusions was also performed to correlate results with tumor genotype. In samples with unidentified drivers, transcriptomic profiles were used to establish a BRAF- or RAS-like molecular subtype based on a gene signature derived from The Cancer Genome Atlas. By using immunohistochemistry, we found co-occurrence of stromal CAFs and senescent TC cells at the tumor invasive front, where deposition of collagen (COL1A1) and expression of lysyl oxidase (LOX) enzyme were also detected, in association with features of local invasion. Concurrent high expression of CAFs and of the senescent TC cells markers, COL1A1 and LOX was confirmed in different TC histotypes in proprietary and public gene sets derived from Gene Expression Omnibus (GEO) repository, and especially in BRAF mutated or BRAF-like tumors. In this study, we show that CAFs and senescent TC cells co-occur in various histotypes of BRAF-driven thyroid tumors and localize at the tumor invasive front.
Keywords: BRAF- and RAS-like signaling; BRAFV600E; CAFs; Thyroid cancer; senescent cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Nikiforov Y.E., Seethala R.R., Tallini G., Baloch Z.W., Basolo F., Thompson L.D., Barletta J.A., Wenig B.M., Al G.A., Kakudo K., et al. Nomenclature Revision for Encapsulated Follicular Variant of Papillary Thyroid Carcinoma: A Paradigm Shift to Reduce Overtreatment of Indolent Tumors. JAMA Oncol. 2016;2:1023–1029. doi: 10.1001/jamaoncol.2016.0386. - DOI - PMC - PubMed
-
- Landa I., Ibrahimpasic T., Boucai L., Sinha R., Knauf J.A., Shah R.H., Dogan S., Ricarte-Filho J.C., Krishnamoorthy G.P., Xu B., et al. Genomic and transcriptomic hallmarks of poorly differentiated and anaplastic thyroid cancers. J. Clin. Investig. 2016;126:1052–1066. doi: 10.1172/JCI85271. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

