Hydroxylamine and Carboxymethoxylamine Can Inhibit Toxoplasma gondii Growth through an Aspartate Aminotransferase-Independent Pathway
- PMID: 31907178
- PMCID: PMC7038254
- DOI: 10.1128/AAC.01889-19
Hydroxylamine and Carboxymethoxylamine Can Inhibit Toxoplasma gondii Growth through an Aspartate Aminotransferase-Independent Pathway
Abstract
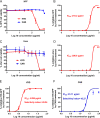
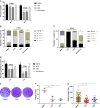
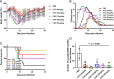
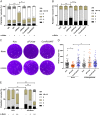
Toxoplasma gondii is an obligate intracellular protozoan parasite and a successful parasitic pathogen in diverse organisms and host cell types. Hydroxylamine (HYD) and carboxymethoxylamine (CAR) have been reported as inhibitors of aspartate aminotransferases (AATs) and interfere with the proliferation in Plasmodium falciparum Therefore, AATs are suggested as drug targets against Plasmodium The T. gondii genome encodes only one predicted AAT in both T. gondii type I strain RH and type II strain PLK. However, the effects of HYD and CAR, as well as their relationship with AAT, on T. gondii remain unclear. In this study, we found that HYD and CAR impaired the lytic cycle of T. gondiiin vitro, including the inhibition of invasion or reinvasion, intracellular replication, and egress. Importantly, HYD and CAR could control acute toxoplasmosis in vivo Further studies showed that HYD and CAR could inhibit the transamination activity of rTgAAT in vitro However, our results confirmed that deficiency of AAT in both RH and PLK did not reduce the virulence in mice, although the growth ability of the parasites was affected in vitro HYD and CAR could still inhibit the growth of AAT-deficient parasites. These findings indicated that HYD and CAR inhibition of T. gondii growth and control of toxoplasmosis can occur in an AAT-independent pathway. Overall, further studies focusing on the elucidation of the mechanism of inhibition are warranted. Our study hints at new substrates of HYD and CAR as potential drug targets to inhibit T. gondii growth.
Keywords: Toxoplasma gondii; aspartate aminotransferase; carboxymethoxylamine; hydroxylamine; toxoplasmosis.
Copyright © 2020 American Society for Microbiology.
Figures



Similar articles
-
Characterization of strain-specific phenotypes associated with knockout of dense granule protein 9 in Toxoplasma gondii.Mol Biochem Parasitol. 2019 Apr;229:53-61. doi: 10.1016/j.molbiopara.2019.01.003. Epub 2019 Mar 5. Mol Biochem Parasitol. 2019. PMID: 30849416
-
Two old drugs, NVP-AEW541 and GSK-J4, repurposed against the Toxoplasma gondii RH strain.Parasit Vectors. 2020 May 11;13(1):242. doi: 10.1186/s13071-020-04094-2. Parasit Vectors. 2020. PMID: 32393321 Free PMC article.
-
Characterization of Plasmodium Atg3-Atg8 Interaction Inhibitors Identifies Novel Alternative Mechanisms of Action in Toxoplasma gondii.Antimicrob Agents Chemother. 2018 Jan 25;62(2):e01489-17. doi: 10.1128/AAC.01489-17. Print 2018 Feb. Antimicrob Agents Chemother. 2018. PMID: 29158278 Free PMC article.
-
Adenosine metabolism in Toxoplasma gondii: potential targets for chemotherapy.Curr Pharm Des. 2007;13(6):581-97. doi: 10.2174/138161207780162836. Curr Pharm Des. 2007. PMID: 17346176 Review.
-
Supply and demand-heme synthesis, salvage and utilization by Apicomplexa.FEBS J. 2021 Jan;288(2):382-404. doi: 10.1111/febs.15445. Epub 2020 Jun 23. FEBS J. 2021. PMID: 32530125 Review.
Cited by
-
Cystathionine-β-Synthase: Molecular Regulation and Pharmacological Inhibition.Biomolecules. 2020 Apr 30;10(5):697. doi: 10.3390/biom10050697. Biomolecules. 2020. PMID: 32365821 Free PMC article. Review.
-
Seroprevalence and Epidemiology of Toxoplasma gondii in Animals in the Qinghai-Tibetan Plateau Area, China.Pathogens. 2021 Apr 6;10(4):432. doi: 10.3390/pathogens10040432. Pathogens. 2021. PMID: 33917355 Free PMC article.
-
Toxoplasma metabolic flexibility in different growth conditions.Trends Parasitol. 2022 Sep;38(9):775-790. doi: 10.1016/j.pt.2022.06.001. Epub 2022 Jun 16. Trends Parasitol. 2022. PMID: 35718642 Free PMC article. Review.
-
PLK:Δgra9 Live Attenuated Strain Induces Protective Immunity Against Acute and Chronic Toxoplasmosis.Front Microbiol. 2021 Mar 11;12:619335. doi: 10.3389/fmicb.2021.619335. eCollection 2021. Front Microbiol. 2021. PMID: 33776955 Free PMC article.
-
Toxoplasma gondii and Neospora caninum Infections in Stray Cats and Dogs in the Qinghai-Tibetan Plateau Area, China.Animals (Basel). 2022 May 28;12(11):1390. doi: 10.3390/ani12111390. Animals (Basel). 2022. PMID: 35681854 Free PMC article.
References
-
- Guo H, Gao Y, Jia H, Moumouni PFA, Masatani T, Liu M, Lee SH, Galon EM, Li J, Li Y, Tumwebaze MA, Benedicto B, Xuan X. 2019. Characterization of strain-specific phenotypes associated with knockout of dense granule protein 9 in Toxoplasma gondii. Mol Biochem Parasitol 229:53–61. doi:10.1016/j.molbiopara.2019.01.003. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

