Regulatory network mediated by RBP-J/NFATc1-miR182 controls inflammatory bone resorption
- PMID: 31908034
- PMCID: PMC7018544
- DOI: 10.1096/fj.201902227R
Regulatory network mediated by RBP-J/NFATc1-miR182 controls inflammatory bone resorption
Abstract
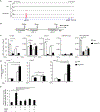
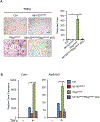
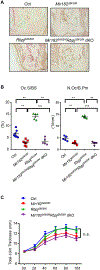
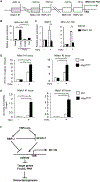
Bone resorption is a severe consequence of inflammatory diseases associated with osteolysis, such as rheumatoid arthritis (RA), often leading to disability in patients. In physiological conditions, the differentiation of bone-resorbing osteoclasts is delicately regulated by the balance between osteoclastogenic and anti-osteoclastogenic mechanisms. Inflammation has complex impact on osteoclastogenesis and bone destruction, and the underlying mechanisms of which, especially feedback inhibition, are underexplored. Here, we identify a novel regulatory network mediated by RBP-J/NFATc1-miR182 in TNF-induced osteoclastogenesis and inflammatory bone resorption. This network includes negative regulator RBP-J and positive regulators, NFATc1 and miR182, of osteoclast differentiation. In this network, miR182 is a direct target of both RBP-J and NFATc1. RBP-J represses, while NFATc1 activates miR182 expression through binding to specific open chromatin regions in the miR182 promoter. Inhibition of miR182 by RBP-J servers as a critical mechanism that limits TNF-induced osteoclast differentiation and inflammatory bone resorption. Inflammation, such as that which occurs in RA, shifts the expression levels of the components in this network mediated by RBP-J/NFATc1-miR182-FoxO3/PKR (previously identified miR182 targets) towards more osteoclastogenic, rather than healthy conditions. Treatment with TNF inhibitors in RA patients reverses the expression changes of the network components and osteoclastogenic potential. Thus, this network controls the balance between activating and repressive signals that determine the extent of osteoclastogenesis. These findings collectively highlight the biological significance and translational implication of this newly identified intrinsic regulatory network in inflammatory osteoclastogenesis and osteolysis.
Keywords: inflammatory bone resorption; osteoclast; osteoimmunology; rheumatoid arthritis.
© 2019 Federation of American Societies for Experimental Biology.
Conflict of interest statement
Figures



References
-
- Novack DV, and Teitelbaum SL (2008) The osteoclast: friend or foe? Annu Rev Pathol 3, 457–484 - PubMed
-
- Schett G, and Teitelbaum SL (2009) Osteoclasts and arthritis. Journal of bone and mineral research : the official journal of the American Society for Bone and Mineral Research 24, 1142–1146 - PubMed
-
- Boyce BF, Yao Z, Zhang Q, Guo R, Lu Y, Schwarz EM, and Xing L (2007) New roles for osteoclasts in bone. Ann N Y Acad Sci 1116, 245–254 - PubMed
-
- Sato K, and Takayanagi H (2006) Osteoclasts, rheumatoid arthritis, and osteoimmunology. Current opinion in rheumatology 18, 419–426 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials
Miscellaneous

