C3 Transferase-Expressing scAAV2 Transduces Ocular Anterior Segment Tissues and Lowers Intraocular Pressure in Mouse and Monkey
- PMID: 31909087
- PMCID: PMC6938898
- DOI: 10.1016/j.omtm.2019.11.017
C3 Transferase-Expressing scAAV2 Transduces Ocular Anterior Segment Tissues and Lowers Intraocular Pressure in Mouse and Monkey
Abstract
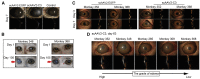
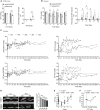
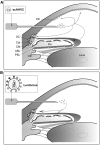
Glaucoma is a lifelong disease with elevated intraocular pressure (IOP) as the main risk factor, and reduction of IOP remains the major treatment for this disease. However, current IOP-lowering therapies are far from being satisfactory. We have demonstrated that the lentivirus-mediated exoenzyme C3 transferase (C3) expression in rat and monkey eyes induced relatively long-term IOP reduction. We now show that intracameral injection of self-complementary AAV2 containing a C3 gene into mouse and monkey eyes resulted in morphological changes in trabecular meshwork and IOP reduction. The vector-transduced corneal endothelium and the C3 transgene expression, not vector itself, induced corneal edema as a result of actin-associated endothelial barrier disruption. There was a positive (quadratic) correlation between measured IOP and grade of corneal edema. This is the first report of using an AAV to transduce the trabecular meshwork of monkeys with a gene capable of altering cellular structure and physiology, indicating a potential gene therapy for glaucoma.
Keywords: C3 transferase; intraocular pressure; monkey; mouse; scAAV2.
© 2019 The Authors.
Figures



Similar articles
-
Evaluation of trabecular meshwork-specific promoters in vitro and in vivo using scAAV2 vectors expressing C3 transferase.Int J Ophthalmol. 2023 Aug 18;16(8):1196-1209. doi: 10.18240/ijo.2023.08.03. eCollection 2023. Int J Ophthalmol. 2023. PMID: 37602341 Free PMC article.
-
Lentiviral Vector-Mediated Expression of Exoenzyme C3 Transferase Lowers Intraocular Pressure in Monkeys.Mol Ther. 2019 Jul 3;27(7):1327-1338. doi: 10.1016/j.ymthe.2019.04.021. Epub 2019 May 9. Mol Ther. 2019. PMID: 31129118 Free PMC article.
-
scAAV2-Mediated Expression of Thioredoxin 2 and C3 Transferase Prevents Retinal Ganglion Cell Death and Lowers Intraocular Pressure in a Mouse Model of Glaucoma.Int J Mol Sci. 2023 Nov 13;24(22):16253. doi: 10.3390/ijms242216253. Int J Mol Sci. 2023. PMID: 38003443 Free PMC article.
-
Serotonin-2 receptor agonists as novel ocular hypotensive agents and their cellular and molecular mechanisms of action.Curr Drug Targets. 2010 Aug;11(8):978-93. doi: 10.2174/138945010791591278. Curr Drug Targets. 2010. PMID: 20426763 Review.
-
Elevation of intraocular pressure in rodents using viral vectors targeting the trabecular meshwork.Exp Eye Res. 2015 Dec;141:33-41. doi: 10.1016/j.exer.2015.04.003. Epub 2015 May 27. Exp Eye Res. 2015. PMID: 26025608 Free PMC article. Review.
Cited by
-
Metformin protects trabecular meshwork against oxidative injury via activating integrin/ROCK signals.Elife. 2023 Jan 4;12:e81198. doi: 10.7554/eLife.81198. Elife. 2023. PMID: 36598818 Free PMC article.
-
The concept of gene therapy for glaucoma: the dream that has not come true yet.Neural Regen Res. 2024 Jan;19(1):92-99. doi: 10.4103/1673-5374.375319. Neural Regen Res. 2024. PMID: 37488850 Free PMC article. Review.
-
Adeno-associated virus vectors for retinal gene therapy in basic research and clinical studies.Front Med (Lausanne). 2023 Dec 1;10:1310050. doi: 10.3389/fmed.2023.1310050. eCollection 2023. Front Med (Lausanne). 2023. PMID: 38105897 Free PMC article. Review.
-
AZD6738 Inhibits fibrotic response of conjunctival fibroblasts by regulating checkpoint kinase 1/P53 and PI3K/AKT pathways.Front Pharmacol. 2022 Sep 20;13:990401. doi: 10.3389/fphar.2022.990401. eCollection 2022. Front Pharmacol. 2022. PMID: 36204234 Free PMC article.
-
A Remote Monitoring System for Rodent Infestation Based on LoRaWAN.Sensors (Basel). 2023 Apr 22;23(9):4185. doi: 10.3390/s23094185. Sensors (Basel). 2023. PMID: 37177388 Free PMC article.
References
-
- Jonas J.B., Aung T., Bourne R.R., Bron A.M., Ritch R., Panda-Jonas S. Glaucoma. Lancet. 2017;390:2183–2193. - PubMed
-
- Ayyala R.S., Chaudhry A.L., Okogbaa C.B., Zurakowski D. Comparison of surgical outcomes between canaloplasty and trabeculectomy at 12 months’ follow-up. Ophthalmology. 2011;118:2427–2433. - PubMed
-
- Inazaki H., Kobayashi S., Anzai Y., Satoh H., Sato S., Inoue M., Yamane S., Kadonosono K. One-year efficacy of adjunctive use of Ripasudil, a rho-kinase inhibitor, in patients with glaucoma inadequately controlled with maximum medical therapy. Graefes Arch. Clin. Exp. Ophthalmol. 2017;255:2009–2015. - PubMed
-
- Toris C.B., McLaughlin M.A., Dworak D.P., Fan S., Havens S., Zhan G.L., Horan N., Prasanna G. Effects of Rho kinase inhibitors on intraocular pressure and aqueous humor dynamics in nonhuman primates and rabbits. J. Ocul. Pharmacol. Ther. 2016;32:355–364. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

