The Role of Electrostatics and Folding Kinetics on the Thermostability of Homologous Cold Shock Proteins
- PMID: 31910002
- PMCID: PMC7450717
- DOI: 10.1021/acs.jcim.9b00797
The Role of Electrostatics and Folding Kinetics on the Thermostability of Homologous Cold Shock Proteins
Abstract
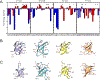
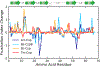
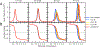
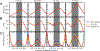
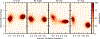
Understanding which aspects contribute to the thermostability of proteins is a challenge that has persisted for decades, and it is of great relevance for protein engineering. Several types of interactions can influence the thermostability of a protein. Among them, the electrostatic interactions have been a target of particular attention. Aiming to explore how this type of interaction can affect protein thermostability, this paper investigated four homologous cold shock proteins from psychrophilic, mesophilic, thermophilic, and hyperthermophilic organisms using a set of theoretical methodologies. It is well-known that electrostatics as well as hydrophobicity are key-elements for the stabilization of these proteins. Therefore, both interactions were initially analyzed in the native structure of each protein. Electrostatic interactions present in the native structures were calculated with the Tanford-Kirkwood model with solvent accessibility, and the amount of hydrophobic surface area buried upon folding was estimated by measuring both folded and extended structures. On the basis of Energy Landscape Theory, the local frustration and the simplified alpha-carbon structure-based model were modeled with a Debye-Hückel potential to take into account the electrostatics and the effects of an implicit solvent. Thermodynamic data for the structure-based model simulations were collected and analyzed using the Weighted Histogram Analysis and Stochastic Diffusion methods. Kinetic quantities including folding times, transition path times, folding routes, and Φ values were also obtained. As a result, we found that the methods are able to qualitatively infer that electrostatic interactions play an important role on the stabilization of the most stable thermophilic cold shock proteins, showing agreement with the experimental data.
Figures

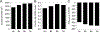
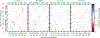

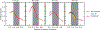
References
-
- Bye J; Platts L; Falconer R Biopharmaceutical Liquid Formulation: A Review of the Science of Protein Stability and Solubility in Aqueous Environments. Biotech. Lett 2014, 36, 869–875. - PubMed
-
- Perl D; Schmid FX Electrostatic Stabilization of a Thermophilic Cold Shock Protein. J. Mol. Biol 2001, 313, 343–357. - PubMed
-
- Jaenicke R; Böhm G. The Stability of Proteins in Extreme Environments. Curr. Opin. Struct. Biol 1998, 8, 738–748. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

