A kinase of many talents: non-neuronal functions of CDK5 in development and disease
- PMID: 31910742
- PMCID: PMC7014686
- DOI: 10.1098/rsob.190287
A kinase of many talents: non-neuronal functions of CDK5 in development and disease
Abstract
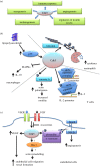
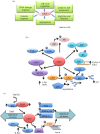
The cyclin-dependent kinase 5 (CDK5) represents an unusual member of the family of cyclin-dependent kinases, which is activated upon binding to non-cyclin p35 and p39 proteins. The role of CDK5 in the nervous system has been very well established. In addition, there is growing evidence that CDK5 is also active in non-neuronal tissues, where it has been postulated to affect a variety of functions such as the immune response, angiogenesis, myogenesis, melanogenesis and regulation of insulin levels. Moreover, high levels of CDK5 have been observed in different tumour types, and CDK5 was proposed to play various roles in the tumorigenic process. In this review, we discuss these various CDK5 functions in normal physiology and disease, and highlight the therapeutic potential of targeting CDK5.
Keywords: CDK5; cancer; cyclin-dependent kinases; development; non-neuronal functions.
Conflict of interest statement
P.S. has served as a consultant at Novartis, Genovis, Guidepoint, The Planning Shop, ORIC Pharmaceuticals and Exo Therapeutics; his laboratory receives research funding from Novartis.
Figures
References
-
- Lewl J. 1992. Purification and characterization of a novel proline-directed protein kinase from bovine brain. J. Biol. Chem. 267, 13 383–13 890. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
