Superior Frontal Sulcus Focal Cortical Dysplasia Type II: An MRI, PET, and Quantified SEEG Study
- PMID: 31920906
- PMCID: PMC6915108
- DOI: 10.3389/fneur.2019.01253
Superior Frontal Sulcus Focal Cortical Dysplasia Type II: An MRI, PET, and Quantified SEEG Study
Abstract
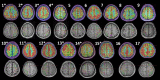
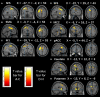
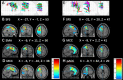
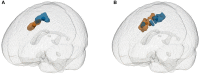
Purpose: The superior frontal sulcus (SFS), located in the prefrontal and premotor cortex, is considered as one of the common locations of focal cortical dysplasia (FCD). However, the characteristics of seizures arising from this area are incompletely known. The primary purpose of this study was to investigate the clinical features and the epileptic networks of seizures originating from the SFS. Methods: We included seventeen patients with type II FCD within the SFS. SFS was identified both visually and automatically. Semiological features were evaluated and grouped. Interictal 18FDG-PET imaging in all patients was compared to controls using statistical parametric mapping (SPM-PET). In those subjects with stereoelectroencephalography (SEEG), two different quantitative intracranial electroencephalography analyses were applied. Finally, the locations of the SFS-related hypometabolic regions and epileptogenic zones (EZs) were transformed into standard space for group analysis. Results: We identified two semiological groups. Group 1 (9/17) showed elementary motor signs (head version and tonic posturing), while group 2 (8/17) exhibited complex motor behavior (fear, hypermotor, and ictal pouting). Based on SPM-PET, an SFS-supplementary motor area (SMA) epileptic propagation network was found in group 1, and an SFS-middle cingulate cortex (MCC)-pregenual anterior cingulate cortex (pACC) propagation network was discovered in group 2. Intracranial EEG analysis suggested similar affected structures with high epileptogenicity. The SFS-related hypometabolic regions and EZs in these groups showed a posterior-anterior spatial relationship. Conclusions: Even though originating from the spatially restricted cortex, SFS seizures can be divided into two groups based on semiological features. The SFS-SMA and SFS-MCC-pACC epileptic propagation networks may play pivotal roles in the generation of different semiologies. The posterior-anterior spatial relationship of both hypometabolic regions and EZs provides potentially useful information for distinguishing different types of SFS seizures and surgical evaluation.
Keywords: epileptic network; epileptogenic zone; focal cortical dysplasia; semiology; superior frontal sulcus.
Copyright © 2019 Zhang, Zhao, McGonigal, Hu, Wang, Shao, Ma, Zhang and Zhang.
Figures

Similar articles
-
Electroclinical features of insulo-opercular epilepsy: an SEEG and PET study.Ann Clin Transl Neurol. 2019 Jul;6(7):1165-1177. doi: 10.1002/acn3.789. Epub 2019 Jun 11. Ann Clin Transl Neurol. 2019. PMID: 31353858 Free PMC article.
-
Electroclinical characteristics of seizures arising from the precuneus based on stereoelectroencephalography (SEEG).BMC Neurol. 2018 Aug 13;18(1):110. doi: 10.1186/s12883-018-1119-z. BMC Neurol. 2018. PMID: 30103717 Free PMC article.
-
Neural networks underlying hyperkinetic seizures: A quantitative PET and SEEG study.Epilepsy Behav. 2021 Sep;122:108130. doi: 10.1016/j.yebeh.2021.108130. Epub 2021 Jun 18. Epilepsy Behav. 2021. PMID: 34153637
-
Emergence of semiology in epileptic seizures.Epilepsy Behav. 2014 Sep;38:94-103. doi: 10.1016/j.yebeh.2013.12.003. Epub 2014 Jan 11. Epilepsy Behav. 2014. PMID: 24424286 Review.
-
Frontal lobe seizures.Neurologist. 2011 May;17(3):125-35. doi: 10.1097/NRL.0b013e31821733db. Neurologist. 2011. PMID: 21532379 Review.
Cited by
-
Long-term seizure outcome with the surgically remediable syndrome of frontal lobe epilepsy associated with superior frontal sulcus-related dysplasia.Front Neurol. 2023 Mar 23;14:1096712. doi: 10.3389/fneur.2023.1096712. eCollection 2023. Front Neurol. 2023. PMID: 37034087 Free PMC article.
-
The superior frontal sulcus in the human brain: Morphology and probability maps.Hum Brain Mapp. 2024 Apr;45(5):e26635. doi: 10.1002/hbm.26635. Hum Brain Mapp. 2024. PMID: 38544425 Free PMC article.
-
Establishment of a normal control model of children's brain 18-fluorodeoxyglucose positron emission tomography and analysis of the changing pattern in patients aged 0-14 years.Quant Imaging Med Surg. 2024 Jul 1;14(7):4703-4713. doi: 10.21037/qims-23-1809. Epub 2024 Jun 27. Quant Imaging Med Surg. 2024. PMID: 39022258 Free PMC article.
-
Symptom network analysis of prefrontal seizures.Epilepsia. 2025 Jul;66(7):2566-2577. doi: 10.1111/epi.18372. Epub 2025 Mar 19. Epilepsia. 2025. PMID: 40105434 Free PMC article.
-
Magnetic resonance-guided laser interstitial thermal therapy vs. stereoelectroencephalography-guided radiofrequency thermocoagulation in epilepsy patients with focal cortical dysplasia: a systematic review and meta-analysis.Front Neurol. 2023 Oct 20;14:1241763. doi: 10.3389/fneur.2023.1241763. eCollection 2023. Front Neurol. 2023. PMID: 37928136 Free PMC article.
References
LinkOut - more resources
Full Text Sources

