Seminal Plasma Modifies the Transcriptional Pattern of the Endometrium and Advances Embryo Development in Pigs
- PMID: 31921921
- PMCID: PMC6930161
- DOI: 10.3389/fvets.2019.00465
Seminal Plasma Modifies the Transcriptional Pattern of the Endometrium and Advances Embryo Development in Pigs
Abstract
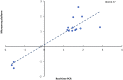
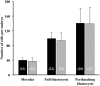
Background: Seminal plasma (SP) promotes sperm survival and fertilizing capacity, and potentially affects embryo development, presumably via specific signaling pathways to the internal female genital tract. Objectives: This study evaluated how heterologous SP, infused immediately before postcervical artificial insemination (AI) affected embryo development and the transcriptional pattern of the pig endometria containing embryos. Materials and Methods: Postweaning estrus sows (n = 34) received 40-mL intrauterine infusions of either heterologous pooled SP or Beltsville Thawing Solution (BTS; control) 30 min before AI of semen extended to 10% of homologous SP. Embryos (all sows) and endometrium samples (3 sows/group) were removed during laparotomy 6 days after the infusion of SP or BTS to morphologically evaluate the embryos to determine their developmental stage and to analyze the endometrial transcriptome using microarrays (PORGENE 1.0 ST GeneChip array, Affymetrix) followed by qPCR for further validation. Results: Embryo viability was equal between the groups (~93%), but embryo development was significantly (P < 0.05) more advanced in the SP-treated group compared to control. A total of 1,604 endometrium transcripts were differentially expressed in the SP group compared to the control group. An enrichment analysis showed an overrepresentation of genes and pathways associated with the immune response, cytokine signaling, cell cycle, cell adhesion, and hormone response, among others. Conclusions: SP infusions prior to AI positively impacted the preimplantation embryo development and altered the expression of the endometrial genes and pathways potentially involved in embryo development.
Keywords: embryo; endometrium; pig; preimplantation; seminal plasma; transcriptome.
Copyright © 2019 Martinez, Cambra, Parrilla, Roca, Ferreira-Dias, Pallares, Lucas, Vazquez, Martinez, Gil, Rodriguez-Martinez, Cuello and Álvarez-Rodriguez.
Figures






Similar articles
-
Seminal Plasma Induces Overexpression of Genes Associated with Embryo Development and Implantation in Day-6 Porcine Blastocysts.Int J Mol Sci. 2020 May 22;21(10):3662. doi: 10.3390/ijms21103662. Int J Mol Sci. 2020. PMID: 32455957 Free PMC article.
-
Intrauterine Infusion of TGF-β1 Prior to Insemination, Alike Seminal Plasma, Influences Endometrial Cytokine Responses but Does Not Impact the Timing of the Progression of Pre-Implantation Pig Embryo Development.Biology (Basel). 2021 Feb 17;10(2):159. doi: 10.3390/biology10020159. Biology (Basel). 2021. PMID: 33671276 Free PMC article.
-
Uterine immune reaction and reproductive performance of sows inseminated with extended semen and infused with pooled whole dead semen.J Anim Sci. 2003 Nov;81(11):2818-25. doi: 10.2527/2003.81112818x. J Anim Sci. 2003. PMID: 14601886
-
Seminal fluid signaling in the female reproductive tract: lessons from rodents and pigs.J Anim Sci. 2007 Mar;85(13 Suppl):E36-44. doi: 10.2527/jas.2006-578. Epub 2006 Nov 3. J Anim Sci. 2007. PMID: 17085725 Review.
-
Influence of semen on inflammatory modulators of embryo implantation.Soc Reprod Fertil Suppl. 2006;62:231-45. Soc Reprod Fertil Suppl. 2006. PMID: 16866321 Review.
Cited by
-
Boar seminal plasma: current insights on its potential role for assisted reproductive technologies in swine.Anim Reprod. 2020 Jul 21;17(3):e20200022. doi: 10.1590/1984-3143-AR2020-0022. Anim Reprod. 2020. PMID: 33029213 Free PMC article. Review.
-
Seminal Plasma: Relevant for Fertility?Int J Mol Sci. 2021 Apr 22;22(9):4368. doi: 10.3390/ijms22094368. Int J Mol Sci. 2021. PMID: 33922047 Free PMC article. Review.
-
Bicarbonate-Triggered In Vitro Capacitation of Boar Spermatozoa Conveys an Increased Relative Abundance of the Canonical Transient Receptor Potential Cation (TRPC) Channels 3, 4, 6 and 7 and of CatSper-γ Subunit mRNA Transcripts.Animals (Basel). 2022 Apr 13;12(8):1012. doi: 10.3390/ani12081012. Animals (Basel). 2022. PMID: 35454259 Free PMC article.
-
Blastocyst-Bearing Sows Display a Dominant Anti-Inflammatory Cytokine Profile Compared to Cyclic Sows at Day 6 of the Cycle.Animals (Basel). 2020 Nov 4;10(11):2028. doi: 10.3390/ani10112028. Animals (Basel). 2020. PMID: 33158034 Free PMC article.
-
Seminal Plasma Modulates miRNA Expression by Sow Genital Tract Lining Explants.Biomolecules. 2020 Jun 19;10(6):933. doi: 10.3390/biom10060933. Biomolecules. 2020. PMID: 32575588 Free PMC article.
References
-
- Rodin IM, Lipatov VI. Artificial insemination of sows. Problemy Zhivotnovodstva. (1935) 9:108–13.
-
- Lyczynski A, Soczywko T, Rillo SM, de Alba Romero C. The effects of Predil-MRA synthetic seminal plasma used to inseminate sows and gilts on their reproductive efficiency. In: Johnson LA, Guthrie HD, editors. Boar Semen Preservation IV. Lawrence, KS: Allen Press, Inc; (2000). p. 250.

