Arterial spin labeling versus 18F-FDG-PET to identify mild cognitive impairment
- PMID: 31931403
- PMCID: PMC6957781
- DOI: 10.1016/j.nicl.2019.102146
Arterial spin labeling versus 18F-FDG-PET to identify mild cognitive impairment
Abstract
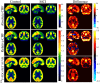
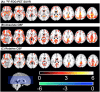
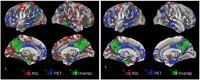
Neurodegenerative biomarkers support diagnosis and measurement of disease progression in the Alzheimer's disease (AD) continuum. 18F-Fluorodeoxyglucose Positron Emission Tomography (18F-FDG-PET), which measures glucose metabolism, is one of the most commonly used biomarkers of neurodegeneration, but is expensive and requires exposure to ionizing radiation. Arterial Spin Labeled (ASL) perfusion Magnetic Resonance Imaging (MRI) provides non invasive quantification of cerebral blood flow (CBF), which is believed to be tightly coupled to glucose metabolism. Here we aimed to compare the performances of ASL derived CBF and 18F-FDG-PET derived standardized uptake value ratio (SUVR) in discriminating patients with mild cognitive impairment (MCI) from older Controls. 2D pseudo continuous ASL and 18F-FDG-PET data with adequate scan quality from 50 MCI study participants (age=73.0 ± 7.0 years, 16 female) and 35 older controls (age=70.2 ± 6.9 years, 20 female), acquired in close temporal proximity, usually on the same day, were considered for this study. We assessed Control-patient group differences both at voxel level and within a priori regions of interest (ROIs). We also compared their area under receiver operating characteristic curves (AUC) with mean CBF or SUVR in a priori selected posterior cingulate cortex (PCC). CBF and 18F-FDG-PET showed abnormalities in similar areas, particularly in medial temporoparietal regions, consistent with the typically observed pattern of prodromal AD. The hypoperfusion pattern with relative CBF (obtained by normalizing voxel CBF values with mean CBF in putamen) was more localized than with absolute CBF. Pearson's correlation coefficients between the T-scores corresponding to the group-differences obtained with 18F-FDG-PET SUVR and absolute and relative ASL CBF were 0.46 and 0.43 (p<0.001), respectively. ROI analyses were also consistent, with the strongest differences observed in PCC (p<0.01). 18F-FDG-PET SUVR, absolute and relative CBF in the PCC ROI demonstrated moderate and similar discriminatory power in predicting MCI status with AUC of 0.71 ± 0.12, 0.77 ± 0.12 and 0.74 ± 0.13, respectively. In conclusion, ASL CBF may be a reasonable, less expensive and safer substitute for 18F-FDG-PET in clinical research.
Keywords: (18)F-Fluorodeoxyglucose positron emission tomography; Alzheimer's disease; Arterial spin labeling; Hypometabolism; Hypoperfusion; Mild cognitive impairment.
Copyright © 2020 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest We believe there is no conflict of interest related to this work. However we disclose here that Dr. Wolk received personal fees for consultation from GE Healthcare, Merck, Eli Lilly, and Jannsen during the conduct of the study; and received grants from Avid Radiopharmaceuticals, Eli Lilly, Merck, Functional Neuromodulation, and Biogen outside the submitted work. Dr. Nasrallah received a grant from Avid Radiopharmaceuticals/Eli Lilly outside the submitted work.
Figures


References
-
- Albert M.S., DeKosky S.T., Dickson D., Dubois B., Feldman H.H., Fox N.C., Gamst A., Holtzman D.M., Jagust W.J., Petersen R.C., Snyder P.J., Carrillo M.C., Thies B., Phelps C.H. The diagnosis of mild cognitive impairment due to alzheimer's disease: recommendations from the national institute on aging-alzheimer's association workgroups on diagnostic guidelines for Alzheimer's disease. Alzheimers Dement. 2011;7:270–279. - PMC - PubMed
-
- Alsop D.C., Detre J.A., Golay X., Gunther M., Hendrikse J., Hernandez-Garcia L., Lu H., Macintosh B.J., Parkes L.M., Smits M., van Osch M.J., Wang D.J., Wong E.C., Zaharchuk G. Recommended implementation of arterial spin-labeled perfusion mri for clinical applications: a consensus of the ismrm perfusion study group and the european consortium for asl in dementia. Magn. Reson. Med. 2015;73:102–116. - PMC - PubMed
-
- Bailly M., Destrieux C., Hommet C., Mondon K., Cottier J.P., Beaufils E., Vierron E., Vercouillie J., Ibazizene M., Voisin T., Payoux P., Barre L., Camus V., Guilloteau D., Ribeiro M.J. Precuneus and cingulate cortex atrophy and hypometabolism in patients with alzheimer's disease and mild cognitive impairment: mri and (18)F-FDG pet quantitative analysis using freesurfer. Biomed. Res. Int. 2015;2015 - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

