Mechanosensitive Ca2+ signaling and coordination is diminished in osteocytes of aged mice during ex vivo tibial loading
- PMID: 31931640
- PMCID: PMC7785095
- DOI: 10.1080/03008207.2020.1712377
Mechanosensitive Ca2+ signaling and coordination is diminished in osteocytes of aged mice during ex vivo tibial loading
Abstract
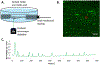
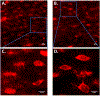
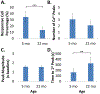
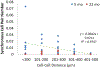
Purpose: The osteocyte is considered the major mechanosensor in bone, capable of detecting forces at a cellular level to coordinate bone formation and resorption. The pathology of age-related bone loss, a hallmark of osteoporosis, is attributed in part to impaired osteocyte mechanosensing. However, real-time evidence of the effect of aging on osteocyte responses to mechanical load is lacking. Intracellular calcium (Ca2+) oscillations have been characterized as an early mechanosensitive response in osteocytes in systems of multiple scales and thus can serve as a real-time measure of osteocyte mechanosensitivity. Our objective was to utilize an ex vivo model to investigate potentially altered mechanosensing in the osteocyte network with aging.Methods: Tibiae were explanted from young-adult (5 mo) and aged (22 mo) female mice and incubated with Fluo-8 AM to visualize osteocyte intracellular Ca2+. Whole tibiae were cyclically loaded while in situ osteocyte Ca2+ dynamics were simultaneously imaged with confocal microscopy. Responsive osteocyte percentage and Ca2+ peak characteristics were quantified, as well as signaling synchrony between paired cells in the field of view.Results: Fewer osteocytes responded to mechanical loading in aged mice compared to young-adult and did so in a delayed manner. Osteocytes from aged mice also lacked the well-correlated relationship between Ca2+ signaling synchrony and cell-cell distance exhibited by young-adult osteocytes.Conclusions: We have demonstrated, for the first time, real-time evidence of the diminished mechanosensing and lack of signaling coordination in aged osteocyte networks in tibial explants, which may contribute to pathology of age-induced bone loss.
Keywords: Ca2+ signaling; Osteocyte; aging; explant; mechanotransduction.
Conflict of interest statement
Disclosure of interest: The authors report no conflict of interest.
Figures
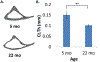

Similar articles
-
In situ intracellular calcium oscillations in osteocytes in intact mouse long bones under dynamic mechanical loading.FASEB J. 2014 Apr;28(4):1582-92. doi: 10.1096/fj.13-237578. Epub 2013 Dec 17. FASEB J. 2014. PMID: 24347610 Free PMC article.
-
Estrogen depletion on In vivo osteocyte calcium signaling responses to mechanical loading.Bone. 2021 Nov;152:116072. doi: 10.1016/j.bone.2021.116072. Epub 2021 Jun 24. Bone. 2021. PMID: 34171514 Free PMC article.
-
Decreased pericellular matrix production and selection for enhanced cell membrane repair may impair osteocyte responses to mechanical loading in the aging skeleton.Aging Cell. 2020 Jan;19(1):e13056. doi: 10.1111/acel.13056. Epub 2019 Nov 19. Aging Cell. 2020. PMID: 31743583 Free PMC article.
-
Osteocyte calcium signaling - A potential translator of mechanical load to mechanobiology.Bone. 2021 Dec;153:116136. doi: 10.1016/j.bone.2021.116136. Epub 2021 Jul 30. Bone. 2021. PMID: 34339908 Review.
-
Aging, Osteocytes, and Mechanotransduction.Curr Osteoporos Rep. 2017 Oct;15(5):401-411. doi: 10.1007/s11914-017-0402-z. Curr Osteoporos Rep. 2017. PMID: 28891009 Free PMC article. Review.
Cited by
-
Culture system for longitudinal monitoring of bone dynamics ex vivo.Biotechnol Bioeng. 2025 Jan;122(1):53-68. doi: 10.1002/bit.28848. Epub 2024 Sep 18. Biotechnol Bioeng. 2025. PMID: 39295202 Free PMC article.
-
P2Y2 Inhibition Modifies the Anabolic Response to Exercise in Adult Mice.Aging Cell. 2025 May;24(5):e14464. doi: 10.1111/acel.14464. Epub 2024 Dec 31. Aging Cell. 2025. PMID: 39741419 Free PMC article.
-
Mitochondrial oxidative stress or decreased autophagy in osteoblast lineage cells is not sufficient to mimic the deleterious effects of aging on bone mechanoresponsiveness.Aging (Albany NY). 2025 Mar 18;17(3):610-629. doi: 10.18632/aging.206213. Epub 2025 Mar 18. Aging (Albany NY). 2025. PMID: 40105873 Free PMC article.
-
Ex vivo Bone Models and Their Potential in Preclinical Evaluation.Curr Osteoporos Rep. 2021 Feb;19(1):75-87. doi: 10.1007/s11914-020-00649-5. Epub 2021 Jan 11. Curr Osteoporos Rep. 2021. PMID: 33428030 Free PMC article. Review.
-
Aging and Altered Gravity: A Cellular Perspective.FASEB J. 2025 Jul 15;39(13):e70777. doi: 10.1096/fj.202402989R. FASEB J. 2025. PMID: 40577073 Free PMC article. Review.
References
-
- Manolagas SC, Jilka RL. Bone marrow, cytokines, and bone remodeling. Emerging insights into the pathophysiology of osteoporosis. N Engl J Med. 1995; 332(5):305–311. - PubMed
-
- Zebaze RM, Ghasem-Zadeh A, Bohte A, Iuliano-Burns S, Mirams M, Price RI, Mackie EJ, Seeman E. Intracortical remodelling and porosity in the distal radius and post-mortem femurs of women: a cross-sectional study. Lancet. 2010; 375(9727):1729–1736. - PubMed
-
- Seeman E, Delmas PD. Bone quality--the material and structural basis of bone strength and fragility. N Engl J Med. 2006; 354(21):2250–2261. - PubMed
-
- Okada S, Yoshida S, Ashrafi SH, Schraufnagel DE. The canalicular structure of compact bone in the rat at different ages. Microsc Microanal. 2002; 8(2):104–115. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous
