Conflicting Actions of Inhalational Anesthetics, Neurotoxicity and Neuroprotection, Mediated by the Unfolded Protein Response
- PMID: 31936788
- PMCID: PMC7013687
- DOI: 10.3390/ijms21020450
Conflicting Actions of Inhalational Anesthetics, Neurotoxicity and Neuroprotection, Mediated by the Unfolded Protein Response
Abstract
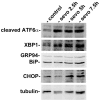
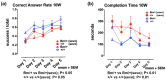
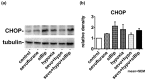
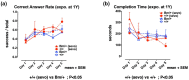
Preclinical studies have shown that exposure of the developing brain to inhalational anesthetics can cause neurotoxicity. However, other studies have claimed that anesthetics can exert neuroprotective effects. We investigated the mechanisms associated with the neurotoxic and neuroprotective effects exerted by inhalational anesthetics. Neuroblastoma cells were exposed to sevoflurane and then cultured in 1% oxygen. We evaluated the expression of proteins related to the unfolded protein response (UPR). Next, we exposed adult mice in which binding immunoglobulin protein (BiP) had been mutated, and wild-type mice, to sevoflurane, and evaluated their cognitive function. We compared our results to those from our previous study in which mice were exposed to sevoflurane at the fetal stage. Pre-exposure to sevoflurane reduced the expression of CHOP in neuroblastoma cells exposed to hypoxia. Anesthetic pre-exposure also significantly improved the cognitive function of adult wild-type mice, but not the mutant mice. In contrast, mice exposed to anesthetics during the fetal stage showed cognitive impairment. Our data indicate that exposure to inhalational anesthetics causes endoplasmic reticulum (ER) stress, and subsequently leads to an adaptive response, the UPR. This response may enhance the capacity of cells to adapt to injuries and improve neuronal function in adult mice, but not in developing mice.
Keywords: ER stress; KDEL receptor; anesthetics; chaperone; endoplasmic reticulum; neuroprotection; neurotoxicity; unfolded protein response.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures
References
-
- Jevtovic-Todorovic V., Hartman R.E., Izumi Y., Benshoff N.D., Dikranian K., Zorumski C.F., Olney J.W., Wozniak D.F. Early exposure to common anesthetic agents causes widespread neurodegeneration in the developing rat brain and persistent learning deficits. J. Neurosci. 2003;23:876–882. doi: 10.1523/JNEUROSCI.23-03-00876.2003. - DOI - PMC - PubMed
-
- Stratmann G., Sall J.W., May L.D., Bell J.S., Magnusson K.R., Rau V., Visrodia K.H., Alvi R.S., Ku B., Lee M.T., et al. Isoflurane differentially affects neurogenesis and long-term neurocognitive function in 60-day-old and 7-day-old rats. Anesthesiology. 2009;110:834–848. doi: 10.1097/ALN.0b013e31819c463d. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

