Next-generation proteasome inhibitor oprozomib enhances sensitivity to doxorubicin in triple-negative breast cancer cells
- PMID: 31938346
- PMCID: PMC6958235
Next-generation proteasome inhibitor oprozomib enhances sensitivity to doxorubicin in triple-negative breast cancer cells
Abstract
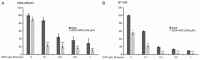
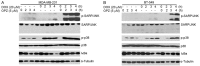
Doxorubicin (DOX) is the most common chemotherapeutic drug for treatment of breast cancer but intrinsic and acquired resistance frequently occurs and severe side effects occur at high doses. DOX might induce activation of NF-κB causing this resistance, in which case proteasome inhibitors could inhibit activation of NF-κB by blocking inhibitory factor κB-alpha degradation. Triple-negative breast cancer (TNBC) is highly progressive and there are no established therapeutic targets against TNBC. Although some proteasome inhibitors have been shown to have antitumor effects in breast cancer, the effect of orally bioavailable proteasome inhibitor oprozomib on TNBC proliferation remains unclear. In the present study, we investigated the role of oprozomib in two TNBC lines, MDA-MB-231 and BT-549. Oprozomib had cytotoxic effects on TNBC cells and increased DOX-induced cytotoxic effects and apoptosis by enhancing DOX-induced JNK/p38 MAPK phosphorylation and inhibiting DOX-induced inhibitory factor êB alpha degradation. These results suggest that oprozomib has potent antitumor effects on TNBC in vitro and can sensitize TNBC cells to DOX treatment. The combination of DOX and oprozomib may be an effective and feasible therapeutic option for TNBC.
Keywords: Proteasome inhibitor; breast cancer; doxorubicin; drug resistance; oprozomib.
IJCEP Copyright © 2018.
Conflict of interest statement
None.
Figures

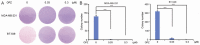


Similar articles
-
Second-generation proteasome inhibitor carfilzomib enhances doxorubicin-induced cytotoxicity and apoptosis in breast cancer cells.Oncotarget. 2016 Nov 8;7(45):73697-73710. doi: 10.18632/oncotarget.12048. Oncotarget. 2016. PMID: 27655642 Free PMC article.
-
A Second-Generation Proteasome Inhibitor and Doxorubicin Modulates IL-6, pSTAT-3 and NF-kB Activity in MDA-MB-231 Breast Cancer Cells.J Nanosci Nanotechnol. 2017 Jan;17(1):175-85. doi: 10.1166/jnn.2017.12427. J Nanosci Nanotechnol. 2017. PMID: 29617099
-
Next-generation proteasome inhibitor MLN9708 sensitizes breast cancer cells to doxorubicin-induced apoptosis.Sci Rep. 2016 May 24;6:26456. doi: 10.1038/srep26456. Sci Rep. 2016. PMID: 27217076 Free PMC article.
-
Apatinib-induced NF-κB inactivation sensitizes triple-negative breast cancer cells to doxorubicin.Am J Transl Res. 2020 Jul 15;12(7):3741-3753. eCollection 2020. Am J Transl Res. 2020. PMID: 32774731 Free PMC article.
-
Advances in Doxorubicin-based nano-drug delivery system in triple negative breast cancer.Front Bioeng Biotechnol. 2023 Nov 17;11:1271420. doi: 10.3389/fbioe.2023.1271420. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 38047286 Free PMC article. Review.
Cited by
-
Anti-proliferative activity of RIHMS-Qi-23 against MCF-7 breast cancer cell line is through inhibition of cell proliferation and senescence but not inhibition of targeted kinases.BMC Cancer. 2023 Nov 2;23(1):1053. doi: 10.1186/s12885-023-11547-1. BMC Cancer. 2023. PMID: 37919708 Free PMC article.
-
Association of Proteasome Activity and Pool Heterogeneity with Markers Determining the Molecular Subtypes of Breast Cancer.Cancers (Basel). 2025 Jan 6;17(1):159. doi: 10.3390/cancers17010159. Cancers (Basel). 2025. PMID: 39796785 Free PMC article.
-
Targeting regulated cell death (RCD) with small-molecule compounds in triple-negative breast cancer: a revisited perspective from molecular mechanisms to targeted therapies.J Hematol Oncol. 2022 Apr 12;15(1):44. doi: 10.1186/s13045-022-01260-0. J Hematol Oncol. 2022. PMID: 35414025 Free PMC article. Review.
-
Mechanisms of doxorubicin-induced drug resistance and drug resistant tumour growth in a murine breast tumour model.BMC Cancer. 2019 Aug 1;19(1):757. doi: 10.1186/s12885-019-5939-z. BMC Cancer. 2019. PMID: 31370818 Free PMC article.
-
CXCR2 Small-Molecule Antagonist Combats Chemoresistance and Enhances Immunotherapy in Triple-Negative Breast Cancer.Front Pharmacol. 2022 Apr 20;13:862125. doi: 10.3389/fphar.2022.862125. eCollection 2022. Front Pharmacol. 2022. PMID: 35517812 Free PMC article.
References
-
- Oberaigner W, Geiger-Gritsch S, Edlinger M, Daniaux M, Knapp R, Hubalek M, Siebert U, Marth C, Buchberger W. Reduction in advanced breast cancer after introduction of a mammography screening program in Tyrol/Austria. Breast. 2017;33:178–82. - PubMed
-
- Huber EM, Groll M. Inhibitors for the immuno- and constitutive proteasome: current and future trends in drug development. Angew Chem Int Ed Engl. 2012;51:8708–20. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous
