Stretch modulation of cardiac contractility: importance of myocyte calcium during the slow force response
- PMID: 31939110
- PMCID: PMC7040129
- DOI: 10.1007/s12551-020-00615-6
Stretch modulation of cardiac contractility: importance of myocyte calcium during the slow force response
Abstract
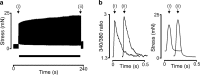
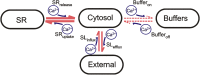
The mechanical response of the heart to myocardial stretch has been understood since the work of muscle physiologists more than 100 years ago, whereby an increase in ventricular chamber filling during diastole increases the subsequent force of contraction. The stretch-induced increase in contraction is biphasic. There is an abrupt increase in the force that coincides with the stretch (the rapid response), which is then followed by a slower response that develops over several minutes (the slow force response, or SFR). The SFR is associated with a progressive increase in the magnitude of the Ca2+ transient, the event that initiates myocyte cross-bridge cycling and force development. However, the mechanisms underlying the stretch-dependent increase in the Ca2+ transient are still debated. This review outlines recent literature on the SFR and summarizes the different stretch-activated Ca2+ entry pathways. The SFR might result from a combination of several different cellular mechanisms initiated in response to activation of different cellular stretch sensors.
Keywords: Autocrine/paracrine response; Calcium influx; Cardiac stretch; G-coupled protein receptors; Slow force response; Stretch-activated channels.
Figures
Similar articles
-
Effect of SR load and pH regulatory mechanisms on stretch-dependent Ca(2+) entry during the slow force response.J Mol Cell Cardiol. 2013 Oct;63:37-46. doi: 10.1016/j.yjmcc.2013.07.008. Epub 2013 Jul 21. J Mol Cell Cardiol. 2013. PMID: 23880608
-
Stretch-activated channels in the heart: contributions to length-dependence and to cardiomyopathy.Prog Biophys Mol Biol. 2008 Jun-Jul;97(2-3):232-49. doi: 10.1016/j.pbiomolbio.2008.02.009. Epub 2008 Feb 13. Prog Biophys Mol Biol. 2008. PMID: 18367238
-
Impact of titin strain on the cardiac slow force response.Prog Biophys Mol Biol. 2017 Nov;130(Pt B):281-287. doi: 10.1016/j.pbiomolbio.2017.06.009. Epub 2017 Jun 22. Prog Biophys Mol Biol. 2017. PMID: 28648628 Free PMC article.
-
The autocrine/paracrine loop after myocardial stretch: mineralocorticoid receptor activation.Curr Cardiol Rev. 2013 Aug;9(3):230-40. doi: 10.2174/1573403x113099990034. Curr Cardiol Rev. 2013. PMID: 23909633 Free PMC article. Review.
-
The slow force response to stretch: Controversy and contradictions.Acta Physiol (Oxf). 2019 May;226(1):e13250. doi: 10.1111/apha.13250. Epub 2019 Jan 25. Acta Physiol (Oxf). 2019. PMID: 30614655 Review.
Cited by
-
Mechano-energetic uncoupling in heart failure.Nat Rev Cardiol. 2025 Jun 22. doi: 10.1038/s41569-025-01167-6. Online ahead of print. Nat Rev Cardiol. 2025. PMID: 40544170 Review.
-
Recent advances in biological pumps as a building block for bioartificial hearts.Front Bioeng Biotechnol. 2023 Jan 20;11:1061622. doi: 10.3389/fbioe.2023.1061622. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 36741765 Free PMC article. Review.
-
Stretch regulation of β2-Adrenoceptor signalling in cardiomyocytes requires caveolae.Cardiovasc Res. 2025 Apr 29;121(3):440-453. doi: 10.1093/cvr/cvae265. Cardiovasc Res. 2025. PMID: 39945052 Free PMC article.
-
Short-term transcriptomic response to plasma membrane injury.Sci Rep. 2021 Sep 27;11(1):19141. doi: 10.1038/s41598-021-98420-y. Sci Rep. 2021. PMID: 34580330 Free PMC article.
-
Harnessing conserved signaling and metabolic pathways to enhance the maturation of functional engineered tissues.NPJ Regen Med. 2022 Sep 3;7(1):44. doi: 10.1038/s41536-022-00246-3. NPJ Regen Med. 2022. PMID: 36057642 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

