Novel Dental Poly (Methyl Methacrylate) Containing Phytoncide for Antifungal Effect and Inhibition of Oral Multispecies Biofilm
- PMID: 31941105
- PMCID: PMC7014161
- DOI: 10.3390/ma13020371
Novel Dental Poly (Methyl Methacrylate) Containing Phytoncide for Antifungal Effect and Inhibition of Oral Multispecies Biofilm
Abstract
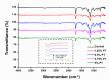
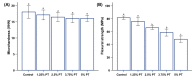
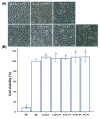
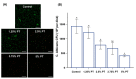
Despite the many advantages of poly (methyl methacrylate) (PMMA) as a dental polymer, its antifungal and antibacterial effects remain limited. Here, phytoncide was incorporated into PMMA to inhibit fungal and biofilm accumulation without impairing the basic and biological properties of PMMA. A variable amount of phytoncide (0 wt % to 5 wt %) was incorporated into PMMA, and the basic material properties of microhardness, flexural strength and gloss were evaluated. In addition, cell viability was confirmed by MTT assay. This MTT assay measures cell viability via metabolic activity, and the color intensity of the formazan correlates viable cells. The fungal adhesion and viability on the PMMA surfaces were evaluated using Candida albicans (a pathogenic yeast). Finally, the thickness of saliva-derived biofilm was estimated. The flexural strength of PMMA decreased with increasing phytoncide contents, whereas there were no significant differences in the microhardness and gloss (p > 0.05) and the cell viability (p > 0.05) between the control and the phytoncide-incorporated PMMA samples. The amounts of adherent Candida albicans colony-forming unit (CFU) counts, and saliva-derived biofilm thickness were significantly lower in the phytoncide-incorporated PMMA compared to the control (p < 0.05). Hence, it was concluded that the incorporation of appropriate amounts of phytoncide in PMMA demonstrated antifungal effects while maintaining the properties, which could be a possible use in dentistry application such as denture base resin.
Keywords: antifungal; dental polymer; oral biofilm; phytoncide; poly (methyl methacrylate).
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Effect of phytochemical-filled microcapsules with antifungal activity on material properties and dimensional accuracy of denture base resin for three-dimensional printing.BMC Oral Health. 2022 May 13;22(1):178. doi: 10.1186/s12903-022-02216-z. BMC Oral Health. 2022. PMID: 35562746 Free PMC article.
-
Evaluation of Physical Properties of Denture Base Resins Containing Silver Nanoparticles of Aloe barbadensis Miller, Morinda citrifolia, and Boesenbergia rotunda and Its Anti-microbial Effect: An In Vitro Study.Cureus. 2023 Nov 4;15(11):e48260. doi: 10.7759/cureus.48260. eCollection 2023 Nov. Cureus. 2023. PMID: 38054116 Free PMC article.
-
Novel Poly(Methyl Methacrylate) Containing Nanodiamond to Improve the Mechanical Properties and Fungal Resistance.Materials (Basel). 2019 Oct 21;12(20):3438. doi: 10.3390/ma12203438. Materials (Basel). 2019. PMID: 31640147 Free PMC article.
-
Incorporation of antimicrobial macromolecules in acrylic denture base resins: a research composition and update.J Prosthodont. 2014 Jun;23(4):284-90. doi: 10.1111/jopr.12105. Epub 2013 Oct 19. J Prosthodont. 2014. PMID: 24138425 Review.
-
Behavior of PMMA Denture Base Materials Containing Titanium Dioxide Nanoparticles: A Literature Review.Int J Biomater. 2019 Jan 17;2019:6190610. doi: 10.1155/2019/6190610. eCollection 2019. Int J Biomater. 2019. PMID: 30792739 Free PMC article. Review.
Cited by
-
Phytochemical-Based Nanoantioxidants Stabilized with Polyvinylpyrrolidone for Enhanced Antibacterial, Antioxidant, and Anti-Inflammatory Activities.Antioxidants (Basel). 2024 Aug 30;13(9):1056. doi: 10.3390/antiox13091056. Antioxidants (Basel). 2024. PMID: 39334715 Free PMC article.
-
Influence of the Manufacturing Method on the Adhesion of Candida albicans and Streptococcus mutans to Oral Splint Resins.Polymers (Basel). 2021 May 11;13(10):1534. doi: 10.3390/polym13101534. Polymers (Basel). 2021. PMID: 34064561 Free PMC article.
-
Synthesis, characterization, and evaluation of quaternary ammonium-based polymerizable antimicrobial monomers for prosthodontic applications.Heliyon. 2022 Aug 23;8(8):e10374. doi: 10.1016/j.heliyon.2022.e10374. eCollection 2022 Aug. Heliyon. 2022. PMID: 36090206 Free PMC article.
-
Surface Characterization, Biocompatibility and Antifungal Efficacy of a Denture-Lining Material Containing Cnidium officinale Extracts.Molecules. 2021 Mar 7;26(5):1440. doi: 10.3390/molecules26051440. Molecules. 2021. PMID: 33799919 Free PMC article.
-
Antifungal Effect of Poly(methyl methacrylate) with Farnesol and Undecylenic Acid against Candida albicans Biofilm Formation.Materials (Basel). 2024 Aug 8;17(16):3936. doi: 10.3390/ma17163936. Materials (Basel). 2024. PMID: 39203113 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources

