Dual-energy spectral CT quantitative parameters for the differentiation of Glioma recurrence from treatment-related changes: a preliminary study
- PMID: 31948400
- PMCID: PMC6966828
- DOI: 10.1186/s12880-019-0406-5
Dual-energy spectral CT quantitative parameters for the differentiation of Glioma recurrence from treatment-related changes: a preliminary study
Abstract
Background: Differentiating glioma recurrence from treatment-related changes can be challenging on conventional imaging. We evaluated the efficacy of quantitative parameters measured by dual-energy spectral computed tomographic (CT) for this differentiation.
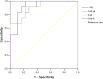
Methods: Twenty-eight patients were examined by dual-energy spectral CT. The effective and normalized atomic number (Zeff and Zeff-N, respectively); spectral Hounsfield unit curve (λHU) slope; and iodine and normalized iodine concentration (IC and ICN, respectively) in the post-treatment enhanced areas were calculated. Pathological results or clinicoradiologic follow-up of ≥2 months were used for final diagnosis. Nonparametric and t-tests were used to compare quantitative parameters between glioma recurrence and treatment-related changes. Sensitivity, specificity, positive and negative predictive values (PPV and NPV, respectively), and accuracy were calculated using receiver operating characteristic (ROC) curves. Predictive probabilities were used to generate ROC curves to determine the diagnostic value.
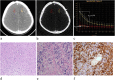
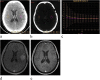
Results: Examination of pre-contrast λHU, Zeff, Zeff-N, IC, ICN, and venous phase ICN showed no significant differences in quantitative parameters (P > 0.05). Venous phase λHU, Zeff, Zeff-N, and IC in glioma recurrence were higher than in treatment-related changes (P < 0.001). The optimal venous phase threshold was 1.03, 7.75, 1.04, and 2.85 mg/cm3, achieving 66.7, 91.7, 83.3, and 91.7% sensitivity; 100.0, 77.8, 88.9, and 77.8% specificity; 100.0, 73.3, 83.3, and 73.3% PPV; 81.8, 93.3, 88.9, and 93.3% NPV; and 86.7, 83.3, 86.7, and 83.3% accuracy, respectively. The respective areas under the curve (AUCs) were 0.912, 0.912, 0.931, and 0.910 in glioma recurrence and treatment-related changes.
Conclusions: Glioma recurrence could be potentially differentiated from treatment-related changes based on quantitative values measured by dual-energy spectral CT imaging.
Keywords: Dual energy spectral CT; Glioma; Recurrence.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

Similar articles
-
Papillary thyroid cancer: dual-energy spectral CT quantitative parameters for preoperative diagnosis of metastasis to the cervical lymph nodes.Radiology. 2015 Apr;275(1):167-76. doi: 10.1148/radiol.14140481. Epub 2014 Dec 17. Radiology. 2015. PMID: 25521777
-
Dual-energy CT quantitative parameters for prediction of prognosis in patients with resectable rectal cancer.Eur Radiol. 2025 Aug;35(8):4945-4956. doi: 10.1007/s00330-025-11398-3. Epub 2025 Feb 8. Eur Radiol. 2025. PMID: 39921716
-
Evaluation of Quantitative Dual-Energy Computed Tomography Parameters for Differentiation of Parotid Gland Tumors.Acad Radiol. 2024 May;31(5):2027-2038. doi: 10.1016/j.acra.2023.08.024. Epub 2023 Sep 18. Acad Radiol. 2024. PMID: 37730491
-
Diagnostic and grading accuracy of 18F-FDOPA PET and PET/CT in patients with gliomas: a systematic review and meta-analysis.BMC Cancer. 2019 Aug 5;19(1):767. doi: 10.1186/s12885-019-5938-0. BMC Cancer. 2019. PMID: 31382920 Free PMC article.
-
Diagnostic accuracy of magnetic resonance diffusion tensor imaging in distinguishing pseudoprogression from glioma recurrence: a systematic review and meta-analysis.Expert Rev Anticancer Ther. 2024 Nov;24(11):1177-1185. doi: 10.1080/14737140.2024.2415404. Epub 2024 Oct 14. Expert Rev Anticancer Ther. 2024. PMID: 39400036
Cited by
-
Quantitative parameter analysis of pretreatment dual-energy computed tomography in nasopharyngeal carcinoma cervical lymph node characteristics and prediction of radiotherapy sensitivity.Radiat Oncol. 2024 Jun 26;19(1):81. doi: 10.1186/s13014-024-02468-9. Radiat Oncol. 2024. PMID: 38918834 Free PMC article.
-
A transferrin receptor targeting dual-modal MR/NIR fluorescent imaging probe for glioblastoma diagnosis.Regen Biomater. 2024 Feb 17;11:rbae015. doi: 10.1093/rb/rbae015. eCollection 2024. Regen Biomater. 2024. PMID: 38487713 Free PMC article.
-
Assessment of the Validity of Carbon Ion Irradiation for C6 Gliomas in Rats.Dose Response. 2025 Apr 15;23(2):15593258251327505. doi: 10.1177/15593258251327505. eCollection 2025 Apr-Jun. Dose Response. 2025. PMID: 40297665 Free PMC article.
-
Utilizing spectral detector computed tomography quantitative parameters via multimodality tumor tracking to predict occult lymph node metastasis in clinical stage 1 pure solid lung adenocarcinoma.J Thorac Dis. 2025 May 30;17(5):2912-2925. doi: 10.21037/jtd-24-1920. Epub 2025 May 23. J Thorac Dis. 2025. PMID: 40529726 Free PMC article.
-
Analysis of material composition and attenuation characteristics of anthropomorphic torso phantoms for dosimetry using dual energy CT technology.Phys Eng Sci Med. 2025 Jun;48(2):675-683. doi: 10.1007/s13246-025-01533-1. Epub 2025 Mar 18. Phys Eng Sci Med. 2025. PMID: 40100568
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

