Activity-Dependent Calcium Signaling in Neurons of the Medial Superior Olive during Late Postnatal Development
- PMID: 31949105
- PMCID: PMC7046326
- DOI: 10.1523/JNEUROSCI.1545-19.2020
Activity-Dependent Calcium Signaling in Neurons of the Medial Superior Olive during Late Postnatal Development
Abstract
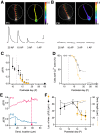
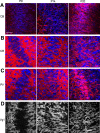
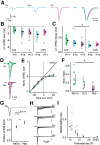
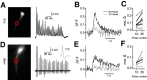
The development of sensory circuits is partially guided by sensory experience. In the medial superior olive (MSO), these refinements generate precise coincidence detection to localize sounds in the azimuthal plane. Glycinergic inhibitory inputs to the MSO, which tune the sensitivity to interaural time differences, undergo substantial structural and functional refinements after hearing onset. Whether excitation and calcium signaling in the MSO are similarly affected by the onset of acoustic experience is unresolved. To assess the time window and mechanism of excitatory and calcium-dependent refinements during late postnatal development, we quantified EPSCs and calcium entry in MSO neurons of Mongolian gerbils of either sex raised in a normal and in an activity altered, omnidirectional white noise environment. Global dendritic calcium transients elicited by action potentials disappeared rapidly after hearing onset. Local synaptic calcium transients decreased, leaving a GluR2 lacking AMPAR-mediated influx as the only activity-dependent source in adulthood. Exposure to omnidirectional white noise accelerated the decrease in calcium entry, leaving membrane properties unaffected. Thus, sound-driven activity accelerates the excitatory refinement and shortens the period of activity-dependent calcium signaling around hearing onset. Together with earlier reports, our findings highlight that excitation, inhibition, and biophysical properties are differentially sensitive to distinct features of sensory experience.SIGNIFICANCE STATEMENT Neurons in the medial superior olive, an ultra-fast coincidence detector for sound source localization, acquire their specialized function through refinements during late postnatal development. The refinement of inhibitory inputs that convey sensitivity to relevant interaural time differences is instructed by the experience of sound localization cues. Which cues instruct the refinement of excitatory inputs, calcium signaling, and biophysical properties is unknown. Here we demonstrate a time window for activity- and calcium-dependent refinements limited to shortly after hearing onset. Exposure to omnidirectional white noise, which suppresses sound localization cues but increases overall activity, accelerates the refinement of calcium signaling and excitatory inputs without affecting biophysical membrane properties. Thus, the refinement of excitation, inhibition, and intrinsic properties is instructed by distinct cues.
Keywords: activity dependence; calcium current; calcium influx; excitatory currents; medial superior olive; postnatal development.
Copyright © 2020 the authors.
Figures


Similar articles
-
Maturation of glycinergic inhibition in the gerbil medial superior olive after hearing onset.J Physiol. 2005 Oct 15;568(Pt 2):497-512. doi: 10.1113/jphysiol.2005.094763. Epub 2005 Aug 11. J Physiol. 2005. PMID: 16096336 Free PMC article.
-
Monaural interaction of excitation and inhibition in the medial superior olive of the mustached bat: an adaptation for biosonar.Proc Natl Acad Sci U S A. 1992 Jun 1;89(11):5108-12. doi: 10.1073/pnas.89.11.5108. Proc Natl Acad Sci U S A. 1992. PMID: 1594619 Free PMC article.
-
Interaural time difference processing in the mammalian medial superior olive: the role of glycinergic inhibition.J Neurosci. 2008 Jul 2;28(27):6914-25. doi: 10.1523/JNEUROSCI.1660-08.2008. J Neurosci. 2008. PMID: 18596166 Free PMC article.
-
The function of the medial superior olive in small mammals: temporal receptive fields in auditory analysis.J Comp Physiol A. 2000 May;186(5):413-23. doi: 10.1007/s003590050441. J Comp Physiol A. 2000. PMID: 10879945 Review.
-
Excitatory action of an immature glycinergic/GABAergic sound localization pathway.Physiol Behav. 2002 Dec;77(4-5):583-7. doi: 10.1016/s0031-9384(02)00905-8. Physiol Behav. 2002. PMID: 12527003 Review.
Cited by
-
Comparative transcriptome analysis provides insights into the molecular mechanisms of high-frequency hearing differences between the sexes of Odorrana tormota.BMC Genomics. 2022 Apr 12;23(1):296. doi: 10.1186/s12864-022-08536-2. BMC Genomics. 2022. PMID: 35410120 Free PMC article.
-
Arrangement of Excitatory Synaptic Inputs on Dendrites of the Medial Superior Olive.J Neurosci. 2021 Jan 13;41(2):269-283. doi: 10.1523/JNEUROSCI.1055-20.2020. Epub 2020 Nov 18. J Neurosci. 2021. PMID: 33208467 Free PMC article.
-
Oligodendrocyte arrangement, identification and morphology in the developing superior olivary complex.Front Cell Neurosci. 2025 Mar 28;19:1561312. doi: 10.3389/fncel.2025.1561312. eCollection 2025. Front Cell Neurosci. 2025. PMID: 40226299 Free PMC article.
-
Periodicity Pitch Perception Part III: Sensibility and Pachinko Volatility.Front Neurosci. 2022 Mar 8;16:736642. doi: 10.3389/fnins.2022.736642. eCollection 2022. Front Neurosci. 2022. PMID: 35356050 Free PMC article.
-
Cellular Strategies for Frequency-Dependent Computation of Interaural Time Difference.Front Synaptic Neurosci. 2022 May 6;14:891740. doi: 10.3389/fnsyn.2022.891740. eCollection 2022. Front Synaptic Neurosci. 2022. PMID: 35602551 Free PMC article. Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
