Mild magnetic nanoparticle hyperthermia enhances the susceptibility of Staphylococcus aureus biofilm to antibiotics
- PMID: 31964196
- PMCID: PMC7730973
- DOI: 10.1080/02656736.2019.1707886
Mild magnetic nanoparticle hyperthermia enhances the susceptibility of Staphylococcus aureus biofilm to antibiotics
Abstract
Objective: A critical challenge in the treatment of biofilm infection is the capacity of biofilm-grown bacteria to develop resistance to traditional antimicrobial therapies. The objective of this study was to validate the therapeutic potential of magnetic nanoparticle/alternating magnetic field (MNP/AMF) hyperthermia in combination with conventional antibiotics against biofilm infection.Materials and methods: The impact of MNP/AMF hyperthermia on the viability of S. aureus biofilm in the absence and presence of antibiotics as well as on the bactericidal activity of macrophages were evaluated at varying conditions of MNPs concentration and AMF intensity using in vitro cell culture models.Results: The application of MNP/AMF alone at a CEM43 thermal dose below the threshold for skin tissue exhibited a modest efficacy in the eradication of Staphylococcus aureus (S. aureus) biofilm (<1-log reduction). The treatment of antibiotics (ciprofloxacin, vancomycin) alone at a bactericidal concentration for planktonic S. aureus had no significant effect on the eradication of biofilm phase of S. aureus. However, when the biofilm was pre-exposed to mild MNP/AMF hyperthermia, the treatment of antibiotics could exhibit bactericidal effects against S. aureus biofilm, which was associated with increased uptake of antibiotics to the bacterial cells. Importantly, the application of MNP/AMF could promote the bactericidal activity of macrophages against intracellular bacteria via MNP-dependent generation of reactive oxygen species (ROS).Conclusion: Our results validate that the application of mild MNP/AMF hyperthermia within a safe thermal dose threshold is synergistic with conventional antibiotics as well as aids host innate immune response of macrophages for the clearance of intracellular bacteria.
Keywords: Magnetic nanoparticles; S. aureus biofilm; alternating magnetic field; antibiotics; hyperthermia.
Conflict of interest statement
Disclosure statement
Joseph Nayfech (Qteris, Inc) declares competing financial interest. Other authors declare no conflict of interest for this study.
Figures



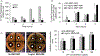
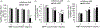

References
-
- Costerton JW, Stewart PS, Greenberg EP. Bacterial biofilms: a common cause of persistent infections. Science. 1999;284(5418): 1318–1322. - PubMed
-
- James GA, Swogger E, Wolcott R, et al. Biofilms in chronic wounds. Wound Repair Regen. 2008;16(1):37–44. - PubMed
-
- Parsek MR, Singh PK. Bacterial biofilms: an emerging link to disease pathogenesis. Annu Rev Microbiol. 2003;57(1):677–701. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
