An Update on the Progress of Isolation, Culture, Storage, and Clinical Application of Human Bone Marrow Mesenchymal Stem/Stromal Cells
- PMID: 31973182
- PMCID: PMC7037097
- DOI: 10.3390/ijms21030708
An Update on the Progress of Isolation, Culture, Storage, and Clinical Application of Human Bone Marrow Mesenchymal Stem/Stromal Cells
Abstract
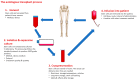
Bone marrow mesenchymal stem/stromal cells (BMSCs), which are known as multipotent cells, are widely used in the treatment of various diseases via their self-renewable, differentiation, and immunomodulatory properties. In-vitro and in-vivo studies have supported the understanding mechanisms, safety, and efficacy of BMSCs therapy in clinical applications. The number of clinical trials in phase I/II is accelerating; however, they are limited in the size of subjects, regulations, and standards for the preparation and transportation and administration of BMSCs, leading to inconsistency in the input and outcome of the therapy. Based on the International Society for Cellular Therapy guidelines, the characterization, isolation, cultivation, differentiation, and applications can be optimized and standardized, which are compliant with good manufacturing practice requirements to produce clinical-grade preparation of BMSCs. This review highlights and updates on the progress of production, as well as provides further challenges in the studies of BMSCs, for the approval of BMSCs widely in clinical application.
Keywords: bone marrow mesenchymal stem cells; clinical application; culture; isolation; stem cell therapy; storage.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Kelly P.N. Skin stem cells regenerate a human epidermis. Science. 2017;358:1267.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

