Endomembrane architecture and dynamics during secretion of the extracellular matrix of the unicellular charophyte, Penium margaritaceum
- PMID: 31974570
- PMCID: PMC7289721
- DOI: 10.1093/jxb/eraa039
Endomembrane architecture and dynamics during secretion of the extracellular matrix of the unicellular charophyte, Penium margaritaceum
Abstract
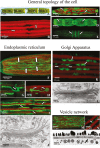
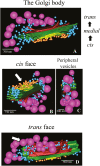
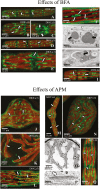
The extracellular matrix (ECM) of many charophytes, the assemblage of green algae that are the sister group to land plants, is complex, produced in large amounts, and has multiple essential functions. An extensive secretory apparatus and endomembrane system are presumably needed to synthesize and secrete the ECM, but structural details of such a system have not been fully characterized. Penium margaritaceum is a valuable unicellular model charophyte for studying secretion dynamics. We report that Penium has a highly organized endomembrane system, consisting of 150-200 non-mobile Golgi bodies that process and package ECM components into different sets of vesicles that traffic to the cortical cytoplasm, where they are transported around the cell by cytoplasmic streaming. At either fixed or transient areas, specific cytoplasmic vesicles fuse with the plasma membrane and secrete their constituents. Extracellular polysaccharide (EPS) production was observed to occur in one location of the Golgi body and sometimes in unique Golgi hybrids. Treatment of cells with brefeldin A caused disruption of the Golgi body, and inhibition of EPS secretion and cell wall expansion. The structure of the endomembrane system in Penium provides mechanistic insights into how extant charophytes generate large quantities of ECM, which in their ancestors facilitated the colonization of land.
Keywords: Penium; Charophyte; Golgi body; endomembrane system; extracellular matrix; tomography.
© The Author(s) 2020. Published by Oxford University Press on behalf of the Society for Experimental Biology.
Figures






Similar articles
-
Endosidin 5 disruption of the Golgi apparatus and extracellular matrix secretion in the unicellular charophyte Penium margaritaceum.Ann Bot. 2023 Jul 10;131(6):967-983. doi: 10.1093/aob/mcad054. Ann Bot. 2023. PMID: 37076269 Free PMC article.
-
Stable transformation and reverse genetic analysis of Penium margaritaceum: a platform for studies of charophyte green algae, the immediate ancestors of land plants.Plant J. 2014 Feb;77(3):339-51. doi: 10.1111/tpj.12375. Epub 2013 Dec 6. Plant J. 2014. PMID: 24308430
-
The cortical cytoskeletal network and cell-wall dynamics in the unicellular charophycean green alga Penium margaritaceum.Ann Bot. 2014 Oct;114(6):1237-49. doi: 10.1093/aob/mcu013. Epub 2014 Mar 5. Ann Bot. 2014. PMID: 24603606 Free PMC article.
-
Penium margaritaceum: A Unicellular Model Organism for Studying Plant Cell Wall Architecture and Dynamics.Plants (Basel). 2014 Nov 18;3(4):543-58. doi: 10.3390/plants3040543. Plants (Basel). 2014. PMID: 27135519 Free PMC article. Review.
-
The extracellular matrix of green algae.Plant Physiol. 2023 Dec 30;194(1):15-32. doi: 10.1093/plphys/kiad384. Plant Physiol. 2023. PMID: 37399237 Free PMC article. Review.
Cited by
-
Conspicuous chloroplast with light harvesting-photosystem I/II megacomplex in marine Prorocentrum cordatum.Plant Physiol. 2024 Apr 30;195(1):306-325. doi: 10.1093/plphys/kiae052. Plant Physiol. 2024. PMID: 38330164 Free PMC article.
-
Role and Evolution of the Extracellular Matrix in the Acquisition of Complex Multicellularity in Eukaryotes: A Macroalgal Perspective.Genes (Basel). 2021 Jul 10;12(7):1059. doi: 10.3390/genes12071059. Genes (Basel). 2021. PMID: 34356075 Free PMC article. Review.
-
Endosidin 5 disruption of the Golgi apparatus and extracellular matrix secretion in the unicellular charophyte Penium margaritaceum.Ann Bot. 2023 Jul 10;131(6):967-983. doi: 10.1093/aob/mcad054. Ann Bot. 2023. PMID: 37076269 Free PMC article.
-
Chemically induced phenotype plasticity in the unicellular zygnematophyte, Penium margaritaceum.Protoplasma. 2024 Nov;261(6):1233-1249. doi: 10.1007/s00709-024-01962-x. Epub 2024 Jul 5. Protoplasma. 2024. PMID: 38967680 Free PMC article.
-
More than a passive barrier: algal cell walls play an active role in determining cell shape, cell size, and organelle morphology.J Exp Bot. 2025 Feb 25;76(4):899-903. doi: 10.1093/jxb/erae411. J Exp Bot. 2025. PMID: 39996292 Free PMC article.
References
-
- Akkerman M, Overdijk EJ, Schel JH, Emons AM, Ketelaar T. 2011. Golgi body motility in the plant cell cortex correlates with actin cytoskeleton organization. Plant & Cell Physiology 52, 1844–1855. - PubMed
-
- Ali O, Traas J. 2016. Force-driven polymerization and turgor-induced wall expansion. Trends in Plant Science 21, 398–409. - PubMed
-
- Barlow LD, Dacks JB. 2018. Seeing the endomembrane system for the trees: evolutionary analysis highlights the importance of plants as models for eukaryotic membrane-trafficking. Seminars in Cell & Developmental Biology 80, 142–152. - PubMed
-
- Bashline L, Lei L, Li S, Gu Y. 2014. Cell wall, cytoskeleton, and cell expansion in higher plants. Molecular Plant 7, 586–600. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

