Real-world Clinical Implementation of Lung Cancer Screening-Evaluating Processes to Improve Screening Guidelines-Concordance
- PMID: 31974902
- PMCID: PMC7174472
- DOI: 10.1007/s11606-019-05539-w
Real-world Clinical Implementation of Lung Cancer Screening-Evaluating Processes to Improve Screening Guidelines-Concordance
Abstract
Background: Lung cancer screening (LCS) requires complex processes to identify eligible patients, provide appropriate follow-up, and manage findings. It is unclear whether LCS in real-world clinical settings will realize the same benefits as the National Lung Screening Trial (NLST).
Objective: To evaluate the impact of process modifications on compliance with LCS guidelines during LCS program implementation, and to compare patient characteristics and outcomes with those in NLST.
Design: Retrospective cohort study.
Setting: Kaiser Permanente Colorado (KPCO), a non-profit integrated healthcare system.
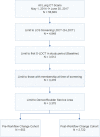
Patients: A total of 3375 patients who underwent a baseline lung cancer screening low-dose computed tomography (S-LDCT) scan between May 2014 and June 2017.
Measurements: Among those receiving an S-LDCT, proportion who met guidelines-based LCS eligibility criteria before and after LCS process modifications, differences in patient characteristics and outcomes between KPCO LCS patients and the NLST cohort, and factors associated with a positive screen.
Results: After modifying LCS eligibility confirmation processes, patients receiving S-LDCT who met guidelines-based LCS eligibility criteria increased from 45.6 to 92.7% (P < 0.001). Prior to changes, patients were older (68 vs. 67 years; P = 0.001), less likely to be current smokers (51.3% vs. 52.5%; P < 0.001), and less likely to have a ≥ 30-pack-year smoking history (50.0% vs. 95.3%; P < 0.001). Compared with NLST participants, KPCO LCS patients were older (67 vs. 60 years; P < 0.001), more likely to currently smoke (52.3% vs. 48.1%; P < 0.001), and more likely to have pulmonary disease. Among those with a positive baseline S-LDCT, the lung cancer detection rate was higher at KPCO (9.4% vs. 3.8%; P < 0.001) and was positively associated with prior pulmonary disease.
Conclusion: Adherence to LCS guidelines requires eligibility confirmation procedures. Among those with a positive baseline S-LDCT, comorbidity burden and lung cancer detection rates were notably higher than in NLST, suggesting that the study of long-term outcomes in patients undergoing LCS in real-world clinical settings is warranted.
Keywords: National Lung Screening Trial; lung cancer screening implementation.
Conflict of interest statement
The authors declare that they do not have a conflict of interest.
Figures
References
-
- Caverly TJ, Fagerlin A, Wiener RS, Slatore CG, Tanner NT, Yun S, et al. Comparison of Observed Harms and Expected Mortality Benefit for Persons in the Veterans Health Affairs Lung Cancer Screening Demonstration Project. JAMA Intern Med. 2018;178(3):426–8. doi: 10.1001/jamainternmed.2017.8170. - DOI - PMC - PubMed
-
- Begnaud A, Hall T, Allen T. Lung Cancer Screening With Low-Dose CT: Implementation Amid Changing Public Policy at One Health Care System. Am Soc Clin Oncol Educ Book. 2016;35:e468–75. doi: 10.14694/edbk_159195. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous