Proteasome Inhibitors for the Treatment of Multiple Myeloma
- PMID: 31979059
- PMCID: PMC7072336
- DOI: 10.3390/cancers12020265
Proteasome Inhibitors for the Treatment of Multiple Myeloma
Abstract
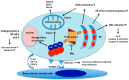
Use of proteasome inhibitors (PIs) has been the therapeutic backbone of myeloma treatment over the past decade. Many PIs are being developed and evaluated in the preclinical and clinical setting. The first-in-class PI, bortezomib, was approved by the US food and drug administration in 2003. Carfilzomib is a next-generation PI, which selectively and irreversibly inhibits proteasome enzymatic activities in a dose-dependent manner. Ixazomib was the first oral PI to be developed and has a robust efficacy and favorable safety profile in patients with multiple myeloma. These PIs, together with other agents, including alkylators, immunomodulatory drugs, and monoclonal antibodies, have been incorporated into several regimens. This review summarizes the biological effects and the results of clinical trials investigating PI-based combination regimens and novel investigational inhibitors and discusses the future perspective in the treatment of multiple myeloma.
Keywords: bortezomib; carfilzomib; ixazomib; multiple myeloma; proteasome inhibitors.
Conflict of interest statement
S.I. received honorarium from Celgene, Bristol-Myers Squibb, Takeda, Ono, and Janssen.
Figures
References
-
- Kumar S.K., Dispenzieri A., Lacy M.Q., Gertz M.A., Buadi F.K., Pandey S., Kapoor P., Dingli D., Hayman S.R., Leung N., et al. Continuous improvement in survival in multiple myeloma: Changes in elderly mortality and outcomes in older patients. Leukemia. 2014;28:1122–1128. doi: 10.1038/leu.2013.313. - DOI - PMC - PubMed
-
- Richardson P.G., Sonneveld P., Schuster M.W., Irwin D., Stadtmauer E.A., Facon T., Harousseau J.L., Ben-Yehuda D., Lonial S., Goldschmidt H., et al. A randomized comparison of bortezomib with high-dose dexamethasone in relapsed multiple myeloma. N. Engl. J. Med. 2005;352:2487–2498. doi: 10.1056/NEJMoa043445. - DOI - PubMed
-
- Herndon T.M., Deisseroth A., Kaminskas E., Kane R.C., Koti K.M., Rothmann M.D., Habtemariam B., Bullock J., Bray J.D., Hawes J., et al. US Food and Drug Administration approval: Carfilzomib for the treatment of multiple myeloma. Clin. Cancer. Res. 2013;19:4559–4563. doi: 10.1158/1078-0432.CCR-13-0755. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous