Hydrogen-exchange kinetics studied through analysis of self-decoupling of nuclear magnetic resonance
- PMID: 31982802
- PMCID: PMC7067644
- DOI: 10.1016/j.jmr.2020.106687
Hydrogen-exchange kinetics studied through analysis of self-decoupling of nuclear magnetic resonance
Abstract
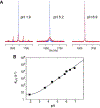
Hydrogen exchange between solute and water molecules occurs across a wide range of timescales. Rapid hydrogen-exchange processes can effectively diminish 1H-15N scalar couplings. We demonstrate that the self-decoupling of 15N nuclear magnetic resonance can allow quantitative investigations of hydrogen exchange on a micro- to millisecond timescale, which is relatively difficult to analyze with other methods. Using a Liouvillian matrix incorporating hydrogen exchange as a mechanism for scalar relaxation, the hydrogen exchange rate can be determined from 15N NMR line shapes recorded with and without 1H decoupling. Self-decoupling offers a simple approach to analyze the kinetics of hydrogen exchange in a wide range of timescale.
Keywords: Hydrogen exchange; Ions; Kinetics; Nuclear magnetic resonance; Self-decoupling.
Copyright © 2020 Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

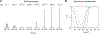
Similar articles
-
Effective strategy to assign ¹H- ¹⁵N heteronuclear correlation NMR signals from lysine side-chain NH3₃⁺ groups of proteins at low temperature.J Biomol NMR. 2014 Sep;60(1):23-7. doi: 10.1007/s10858-014-9854-y. Epub 2014 Aug 17. J Biomol NMR. 2014. PMID: 25129623 Free PMC article.
-
31P NMR probes of chemical dynamics: paramagnetic relaxation enhancement of the (1)H and (31)P NMR resonances of methyl phosphite and methylethyl phosphate anions by selected metal complexes.Inorg Chem. 2001 Dec 17;40(26):6547-54. doi: 10.1021/ic010728w. Inorg Chem. 2001. PMID: 11735462
-
Detecting Bound Ions in Ion Channels by Solid-State NMR Experiments on 15N-Labelled Ammonium Ions.Methods Mol Biol. 2024;2796:23-34. doi: 10.1007/978-1-0716-3818-7_2. Methods Mol Biol. 2024. PMID: 38856893
-
Nuclear magnetic resonance studies of biologically relevant isotopes other than hydrogen.CRC Crit Rev Biochem. 1978;5(1):25-43. doi: 10.3109/10409237809177139. CRC Crit Rev Biochem. 1978. PMID: 357083 Review. No abstract available.
-
Measurement of homo- and heteronuclear J couplings from quantitative J correlation.Methods Enzymol. 1994;239:79-105. doi: 10.1016/s0076-6879(94)39004-5. Methods Enzymol. 1994. PMID: 7830604 Review. No abstract available.
References
-
- Englander SW, Downer NW, Teitelbaum H, Hydrogen exchange, Annu Rev Biochem, 41 (1972) 903–924. - PubMed
-
- Liepinsh E, Otting G, Proton exchange rates from amino acid side chains--implications for image contrast, Magn Reson Med, 35 (1996) 30–42. - PubMed
-
- Englander SW, Kallenbach NR, Hydrogen exchange and structural dynamics of proteins and nucleic acids, Q Rev Biophys, 16 (1983) 521–655. - PubMed
-
- Wales TE, Engen JR, Hydrogen exchange mass spectrometry for the analysis of protein dynamics, Mass Spectrom Rev, 25 (2006) 158–170. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

