Ibuprofen for the prevention of patent ductus arteriosus in preterm and/or low birth weight infants
- PMID: 31985838
- PMCID: PMC6984616
- DOI: 10.1002/14651858.CD004213.pub5
Ibuprofen for the prevention of patent ductus arteriosus in preterm and/or low birth weight infants
Abstract
Background: Patent ductus arteriosus (PDA) complicates the clinical course of preterm infants and increases the risk of adverse outcomes. Indomethacin has been the standard treatment to close a PDA but is associated with renal, gastrointestinal, and cerebral side effects. Ibuprofen has less effect on blood flow velocity to important organs.
Objectives: Primary objectives To determine the effectiveness and safety of ibuprofen compared to placebo/no intervention, or other cyclo-oxygenase inhibitor drugs in the prevention of PDA in preterm infants.
Search methods: We used the standard search strategy of Cochrane Neonatal to search the Cochrane Central Register of Controlled Trials (CENTRAL; 2018, Issue 10), MEDLINE via PubMed (1966 to 17 October 2018), Embase (1980 to 17 October 2018), and CINAHL; 1982 to 17 October 2018). We searched clinical trials databases, conference proceedings, and the reference lists of retrieved articles for randomised controlled trials and quasi-randomised trials.
Selection criteria: Randomised and quasi-randomised controlled trials comparing ibuprofen with placebo/no intervention or other cyclo-oxygenase inhibitor drugs to prevent PDA in preterm or low birth weight infants.
Data collection and analysis: We extracted outcomes data including presence of PDA on day three or four of life (after 72 hours of treatment), need for surgical ligation or rescue treatment with cyclo-oxygenase inhibitors, mortality, cerebral, renal, pulmonary, and gastrointestinal complications. We performed meta-analyses and reported treatment estimates as typical mean difference (MD), risk ratio (RR), risk difference (RD) and, if statistically significant, number needed to treat to benefit (NNTB) or to harm (NNTH), along with their 95% confidence intervals (CI). We assessed between-study heterogeneity by the I-squared test (I²). We used the GRADE approach to assess the quality of evidence.
Main results: In this updated analysis, we included nine trials (N = 1070 infants) comparing prophylactic ibuprofen (IV or oral) with placebo/no intervention or indomethacin. Ibuprofen (IV or oral) probably decreases the risk of PDA on day 3 or 4 (typical RR 0.39, 95% CI 0.31 to 0.48; typical RD -0.26, 95% CI -0.31 to -0.21; NNTB 4, 95% CI 3 to 5; 9 trials; N = 1029) (moderate-quality evidence). In the control group, the spontaneous closure rate was 58% by day 3 to 4 of age. In addition, ibuprofen probably decreases the need for rescue treatment with cyclo-oxygenase inhibitors (typical RR 0.17, 95% CI 0.11 to 0.26; typical RD -0.27, 95% CI -0.32 to -0.22; NNTB 4; 95% CI 3 to 5),and the need for surgical ductal ligation (typical RR 0.46, 95% CI 0.22 to 0.96; typical RD -0.03, 95% CI -0.05 to -0.00; NNTB 33, 95% CI 20 to infinity; 7 trials; N = 925) (moderate-quality evidence). There was a possible decrease in the risk of grade 3 or 4 intraventricular haemorrhage (IVH) in infants receiving prophylactic ibuprofen (typical RR 0.67, 95% CI 0.45 to 1.00; I² = 34%; typical RD -0.04, 95% CI -0.08 to- 0.00; I² = 60%; 7 trials; N = 925) (moderate-quality evidence). High quality evidence showed increased risk for oliguria (typical RR 1.45, 95% CI 1.04 to 2.02; typical RD 0.06, 95% CI 0.01 to 0.11; NNTH 17, 95% CI 9 to 100; 4 trials; N = 747). Low quality results from four studies (N = 202) showed that administering oral ibuprofen may decrease the risk of PDA (typical RR 0.47, 95% CI 0.30 to 0.74) and may increase risk of gastrointestinal bleeding (NNTH 7, 95% CI 4 to 25). No evidence of a difference was identified for mortality, any intraventricular haemorrhage (IVH), or chronic lung disease.
Authors' conclusions: This review shows that prophylactic use of ibuprofen, compared to placebo or no intervention, probably decreases the incidence of patent ductus arteriosus, the need for rescue treatment with cyclo-oxygenase inhibitors, and for surgical ductal closure. Adverse effects associated with ibuprofen (IV or oral) included increased risks for oliguria, increase in serum creatinine levels, and increased risk of gastrointestinal haemorrhage. There was a reduced risk for intraventricular haemorrhage (grade III - IV) but no evidence of a difference in mortality, chronic lung disease, necrotising enterocolitis, or time to reach full feeds. In the control group, the patent ductus arteriosus had closed spontaneously by day 3 or 4 in 58% of neonates. Prophylactic treatment exposes a large proportion of infants unnecessarily to a drug that has important side effects without conferring any important short-term benefits. Current evidence does not support the use of ibuprofen for prevention of patent ductus arteriosus. Until long-term follow-up results of the trials included in this review have been published, no further trials of prophylactic ibuprofen are recommended. A new approach to patent ductus arteriosus management is an early targeted treatment based on echocardiographic criteria within the first 72 hours of life, that have a high sensitivity for diagnosing a patent ductus arteriosus that is unlikely to close spontaneously. Such trials are currently ongoing in many parts of the world. Results of such trials will be included in updates of our "Ibuprofen for treatment of PDA" review.
Contexte: La persistance du canal artériel (PCA) complique l'évolution clinique des prématurés et augmente le risque d'effets indésirables. L'indométhacine a été le traitement standard pour fermer une PCA, mais elle est associée à des effets secondaires rénaux, gastro‐intestinaux et cérébraux. L'ibuprofène a moins d'effet sur la vitesse du flux sanguin vers les organes importants.
Objectifs: Objectifs principaux Déterminer l'efficacité et l'innocuité de l'ibuprofène par rapport au placebo/aucune intervention ou à d'autres inhibiteurs de la cyclo‐oxygénase dans la prévention de la PCA chez les prématurés. STRATÉGIE DE RECHERCHE DOCUMENTAIRE: Nous avons utilisé la stratégie de recherche standard de Cochrane Neonatal pour faire des recherches dans le Registre central des essais contrôlés de Cochrane (CENTRAL ; 2018, numéro 10), MEDLINE via PubMed (1966 au 17 octobre 2018), Embase (1980 au 17 octobre 2018) et CINAHL ; 1982 au 17 octobre 2018). Nous avons également effectué des recherches dans les bases de données d'essais cliniques, les actes de conférence, et les références bibliographiques d'articles sélectionnés pour identifier des essais contrôlés randomisés ou quasi randomisés. CRITÈRES DE SÉLECTION: Essais contrôlés randomisés et quasi‐randomisés comparant l'ibuprofène à un placebo/aucune intervention ou à d'autres inhibiteurs de la cyclo‐oxygénase pour prévenir les PCA chez les prématurés ou les nourrissons de faible poids à la naissance. RECUEIL ET ANALYSE DES DONNÉES: Nous avons extrait des données sur les critères de jugement, y compris la présence de PCA au troisième ou quatrième jour de vie (après 72 heures de traitement), la nécessité d'une ligature chirurgicale ou d'un traitement de secours avec des inhibiteurs de cyclo‐oxygénase, la mortalité, les complications cérébrales, rénales, pulmonaires et gastro‐intestinales. Nous avons effectué des méta‐analyses et reporté les estimations de traitement sous forme de différence moyenne typique (DM), de risque relatif (RR), de différence de risque (DR) et, si elles sont statistiquement significatives, de nombre de sujets à traiter pour en retirer des bénéfices (NNTB) ou des préjudices (NNTH), ainsi que leurs intervalles de confiance (IC) à 95 %. Nous avons évalué l'hétérogénéité entre les études par le test du carré I (I²). Nous avons utilisé l'approche GRADE pour évaluer la qualité des données probantes. RÉSULTATS PRINCIPAUX: Dans cette analyse mise à jour, nous avons inclus neuf essais (N = 1070 nourrissons) comparant l'ibuprofène prophylactique (IV ou oral) avec placebo/aucune intervention ou l’indométhacine. L'ibuprofène (IV ou oral) diminue probablement le risque de PCA au jour 3 ou 4 (RR typique 0,39, IC à 95 % 0,31 à 0,48 ; DR typique ‐0,26, IC à 95 % ‐0,31 à ‐0,21 ; NNTB 4, IC à 95 % 3 à 5 ; 9 essais ; N = 1029) (données probantes de qualité moyenne). Dans le groupe témoin, le taux de fermeture spontanée était de 58 % au jour 3 ou 4. De plus, l'ibuprofène diminue probablement le besoin d'un traitement de secours avec des inhibiteurs de cyclo‐oxygénase (RR typique 0,17, IC à 95 % 0,11 à 0,26 ; DR typique ‐0,27, IC à 95 % ‐0,32 à ‐0,22 ; NSTB 4 ; IC à 95 % 3 à 5),et le besoin de ligature chirurgicale des canaux (RR typique 0,46, IC à 95 % 0,22 à 0,96 ; DR typique ‐0,03, IC à 95 % 0,05 à ‐0,00 ; NNTB 33, IC à 95 % 20 à infini ; 7 essais ; N = 925) (données probantes de qualité moyenne). On a observé une diminution possible du risque d'hémorragie intraventriculaire (HIV) de grade 3 ou 4 chez les nourrissons recevant de l'ibuprofène prophylactique (RR typique 0,67, IC à 95 % 0,45 à 1,00 ; I² = 34 % ; DR typique ‐0,04, IC à 95 % ‐0,08 à 0,00 ; I² = 60 % ; 7 essais ; N = 925) (données probantes de qualité moyenne). Des données probantes de haute qualité ont montré un risque accru d'oligurie (RR typique 1,45, IC à 95 % 1,04 à 2,02 ; DR typique 0,06, IC à 95 % 0,01 à 0,11 ; NNTH 17, IC à 95 % 9 à 100 ; 4 essais ; N = 747). Les résultats de faible qualité de quatre études (N = 202) ont montré que l'administration orale d'ibuprofène peut diminuer le risque de PCA (RR typique 0,47, IC à 95 % 0,30 à 0,74) et peut augmenter le risque de saignement gastro‐intestinal (NNTH 7, IC à 95 % 4 à 25). Aucune donnée probante montrant une différence n'a été relevée en ce qui concerne la mortalité, les hémorragies intraventriculaires (HIV) ou les maladies pulmonaires chroniques.
Conclusions des auteurs: Cette revue montre que l'utilisation prophylactique de l'ibuprofène, comparativement au placebo ou à l'absence d'intervention, diminue probablement la persistance du canal artériel, la nécessité d'un traitement de secours avec des inhibiteurs de cyclo‐oxygénase et d'une fermeture chirurgicale du canal. Les effets indésirables associés à l'ibuprofène (par voie intraveineuse ou orale) comprenaient un risque accru d'oligurie, une augmentation des taux sériques de créatinine et un risque accru d'hémorragie gastro‐intestinale. Le risque d'hémorragie intraventriculaire (grade III ‐ IV) était réduit, mais il n'y avait aucun signe de différence dans la mortalité, les maladies pulmonaires chroniques, l'entérocolite nécrosante ou le délai pour se nourrir complètement. Dans le groupe témoin, la persistance du canal artériel s'était fermée spontanément au jour 3 ou 4 chez 58 % des nouveau‐nés. Le traitement prophylactique expose inutilement une grande proportion de nourrissons à un médicament qui a des effets secondaires importants sans leur procurer d'avantages importants à court terme. Les données probantes actuelles n'appuient pas l'utilisation de l'ibuprofène pour la prévention de la persistance du canal artériel. Jusqu'à ce que les résultats du suivi à long terme des essais inclus dans cette revue aient été publiés, aucun autre essai sur l'ibuprofène prophylactique n'est recommandé. Une nouvelle approche de la prise en charge de la persistance du canal artériel est un traitement précoce ciblé basé sur des critères échocardiographiques dans les 72 premières heures de la vie, qui présentent une sensibilité élevée pour le diagnostic d'une persistance du canal artériel dont la fermeture spontanée est peu probable. De tels essais sont actuellement en cours dans de nombreuses régions du monde. Les résultats de ces essais seront inclus dans les mises à jour de notre revue "Ibuprofen pour le traitement de la PCA".
Antecedentes: El conducto arterioso persistente (CAP) complica el curso clínico de los lactantes prematuros y aumenta el riesgo de resultados adversos. La indometacina ha sido el tratamiento estándar para cerrar el CAP, pero se asocia con efectos secundarios renales, gastrointestinales y cerebrales. El ibuprofeno tiene menos efectos sobre la velocidad del flujo sanguíneo a los órganos importantes.
Objetivos: Objetivos primarios Determinar la efectividad y seguridad del ibuprofeno en comparación con el placebo, con la no intervención o con otros fármacos inhibidores de la ciclooxigenasa en la prevención del CAP en recién nacidos prematuros. MÉTODOS DE BÚSQUEDA: Se utilizó la estrategia de búsqueda estándar del Grupo Cochrane de Neonatología para buscar en el Registro Cochrane Central de Ensayos Controlados (CENTRAL 2018, número 10), MEDLINE vía PubMed (1966 hasta 17 octubre 2018), EMBASE (1980 hasta 17 octubre 2018) y en CINAHL (1982 hasta 17 octubre 2018). También se buscaron ensayos controlados aleatorizados y ensayos cuasialeatorizados en bases de datos de ensayos clínicos, actas de congresos y listas de referencias de los artículos recuperados. CRITERIOS DE SELECCIÓN: Ensayos controlados aleatorizados o cuasialeatorizados que compararon ibuprofeno con placebo o ninguna intervención o con otros fármacos inhibidores de la ciclooxigenasa para la prevención del CAP en lactantes prematuros o de bajo peso al nacer. OBTENCIÓN Y ANÁLISIS DE LOS DATOS: Se extrajeron datos de resultado, como la presencia del CAP al tercer o cuarto día de vida (después de 72 horas de tratamiento), la necesidad de ligadura quirúrgica o el tratamiento de rescate con inhibidores de la ciclooxigenasa, la mortalidad y las complicaciones cerebrales, renales, pulmonares y gastrointestinales. Se realizaron los metanálisis e informaron los cálculos de tratamiento como la diferencia de medias (DM) típica, los riesgos relativos (RR), la diferencia de riesgos (DR) y, si era estadísticamente significativo, el número necesario a tratar para lograr un beneficio (NNTB) o daño (NNTH), junto con los intervalos de confianza (IC) del 95%. Se evaluó la heterogeneidad entre estudios con la prueba de I cuadrado (I²). Se utilizaron los criterios GRADE para evaluar la calidad de la evidencia.
Resultados principales: En este análisis actualizado, se incluyeron nueve ensayos (N = 1070 neonatos) que compararon el ibuprofeno profiláctico (i.v. u oral) con placebo o ninguna intervención o indometacina. El ibuprofeno (intravenoso u oral) probablemente disminuye el riesgo de CAP en el día 3 ó 4 (RR típico 0,39; IC del 95%: 0,31 a 0,48; DR típica ‐0,26; IC del 95%: ‐0,31 a ‐0,21; NNTB 4; IC del 95%: 3 a 5; nueve ensayos; N = 1029) (evidencia de calidad moderada). En el grupo de control, la tasa de cierre espontáneo fue de un 58% a los tres o cuatro días de vida. Además, el ibuprofeno probablemente disminuye la necesidad de tratamiento de rescate con inhibidores de la ciclooxigenasa (RR típico 0,17; IC del 95%: 0,11 a 0,26; DR típica ‐0,27; IC del 95%: ‐0,32 a ‐0).22; NNTB 4; IC del 95%: 3 a 5), y la necesidad de ligadura ductal quirúrgica (RR típico 0,46; IC del 95%: 0,22 a 0,96; DR típica ‐0,03; IC del 95%: ‐0,05 a ‐0,00; NNTB 33; IC del 95%: 20 a infinito; siete ensayos; N = 925) (evidencia de calidad moderada). Hubo una posible disminución en el riesgo de hemorragia intraventricular (Hiv) de grado 3 o 4 en los lactantes que recibieron ibuprofeno profiláctico (RR típico 0,67; IC del 95%: 0,45 a 1,00; I² = 34%; DR típica ‐0,04; IC del 95%: ‐0,08 a 0,00; I² = 60%; siete ensayos; N = 925) (evidencia de calidad moderada). La evidencia de alta calidad mostró un mayor riesgo de oliguria (RR típico 1,45; IC del 95%: 1,04 a 2,02; DR típica 0,06; IC del 95%: 0,01 a 0,11; NNTD 17; IC del 95%: 9 a 100; cuatro ensayos; N = 747). Los resultados de calidad baja de cuatro estudios (N = 202) demostraron que la administración de ibuprofeno oral puede reducir el riesgo de CAP (RR típico 0,47; IC del 95%: 0,30 a 0,74) y puede aumentar el riesgo de hemorragia gastrointestinal (NNTH 7; IC del 95%: 4 a 25). No se identificó evidencia de una diferencia en cuanto a la mortalidad, cualquier grado de hemorragia intraventricular (Hiv) o la enfermedad pulmonar crónica.
Conclusiones de los autores: Esta revisión demuestra que el uso profiláctico de ibuprofeno, en comparación con placebo o ninguna intervención, probablemente disminuye la incidencia de conducto arterioso persistente, la necesidad de tratamiento de rescate con inhibidores de la ciclooxigenasa y de cierre ductal quirúrgico. Los efectos adversos asociados con el ibuprofeno (i.v. u oral) incluyeron mayor riesgo de oliguria, aumento de los niveles de creatinina sérica y mayor riesgo de hemorragia gastrointestinal. Se redujo el riesgo de hemorragia intraventricular (grado III ‐ IV), pero no hubo evidencia de una diferencia en la mortalidad, la enfermedad pulmonar crónica, la enterocolitis necrosante ni el tiempo hasta la alimentación enteral total. En el grupo de control, el conducto arterioso persistente se había cerrado espontáneamente al tercer o cuarto día de vida en un 58% de recién nacidos. El tratamiento profiláctico expone innecesariamente a un gran número de lactantes a un fármaco que tiene efectos secundarios importantes, sin ofrecer efectos beneficiosos importantes a corto plazo. La evidencia actual no apoya el uso del ibuprofeno para la prevención del conducto arterioso persistente. Hasta que se publiquen resultados del seguimiento a largo plazo de los ensayos incluidos en esta revisión, no se recomiendan ensayos adicionales de ibuprofeno profiláctico. Un nuevo enfoque terapéutico para el conducto arterioso persistente consiste en un tratamiento específico inicial sobre la base de criterios ecocardiográficos en las primeras 72 horas de vida, que tienen una sensibilidad alta para diagnosticar un conducto arterioso permeable que tiene poca probabilidad de cerrarse espontáneamente. Estos ensayos están actualmente en curso en muchos países. Los resultados de estos ensayos se incluirán en las actualizaciones de la revisión "Ibuprofeno para el tratamiento del CAP".
Copyright © 2020 The Cochrane Collaboration. Published by John Wiley & Sons, Ltd.
Conflict of interest statement
AO has no interests to declare.
SS has no interests to declare.
Figures


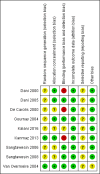
















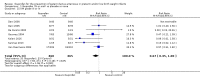




























Update of
-
Ibuprofen for the prevention of patent ductus arteriosus in preterm and/or low birth weight infants.Cochrane Database Syst Rev. 2019 Jun 21;6(6):CD004213. doi: 10.1002/14651858.CD004213.pub4. Cochrane Database Syst Rev. 2019. Update in: Cochrane Database Syst Rev. 2020 Jan 27;1:CD004213. doi: 10.1002/14651858.CD004213.pub5. PMID: 31222841 Free PMC article. Updated.
References
References to studies included in this review
Dani 2000 {published data only}
-
- Dani C, Bertini G, Reali MF, Murru P, Fabris C, Vangi V, et al. Prophylaxis of patent ductus arteriosus with ibuprofen in preterm infants. Acta Paediatrica 2000;89(11):1369‐74. [PUBMED: 11106052] - PubMed
-
- Rubaltelli FF, Bertini G, Reali MF, Vangi V, Dani C. Does early closure of PDA with ibuprofen reduce the severity of RDS in premature infants?. Pediatric Research 1998;43:296. [DOI: 10.1203/00006450-199804001-01758] - DOI
Dani 2005 {published data only}
De Carolis 2000 {published data only}
-
- Carolis MP, Romagnoli C, Polimeni V, Piersigilli F, Zecca E, Papacci P, et al. Prophylactic ibuprofen therapy of patent ductus arteriosus in preterm infants. European Journal of Pediatrics 2000;159(5):364‐8. [PUBMED: 10834523] - PubMed
Gournay 2004 {published data only}
Kalani 2016 {published data only}
-
- Kalani M, Shariat M, Khalesi N, Farahani Z, Ahmadi S. A comparison of early ibuprofen and indomethacin administration to prevent intraventricular hemorrhage among preterm infants. Acta Medica Iranica 2016;54(12):788‐92. [PUBMED: 28120591] - PubMed
Kanmaz 2013 {published data only}
-
- Kanmaz G, Erdeve O, Canpolat FE, Oğuz SS, Uras N, Altug N, et al. Serum ibuprofen levels of extremely preterm infants treated prophylactically with oral ibuprofen to prevent patent ductus arteriosus. European Journal of Clinical Pharmacology 2013;69(5):1075‐81. [DOI: 10.1007/s00228-012-1438-8; PUBMED: 23128963] - DOI - PubMed
Sangtawesin 2006 {published data only}
-
- Sangtawesin V, Sangtawesin C, Raksasinborisut C, Sathirakul K, Kanjanapattanakul W, Khorana M, et al. Oral ibuprofen prophylaxis for symptomatic patent ductus arteriosus of prematurity. Journal of the Medical Association of Thailand 2006;89(3):314‐20. [PUBMED: 16696414] - PubMed
Sangtawesin 2008 {published data only}
-
- Sangtawesin C, Sangtawesin V, Lertsutthiwong W, Kanjanapattanakul W, Khorana M, Ayudhaya JK. Prophylaxis of symptomatic patent ductus arteriosus with oral ibuprofen in very low birth weight infants. Journal of the Medical Association of Thailand 2008;91:S28‐34. [PUBMED: 19255990] - PubMed
Van Overmeire 2004 {published data only}
-
- Overmeire B, Casaer A, Allegaert K, Debauche C, Decaluwe W, Jespers A. Multicenter ibuprofen prophylaxis study (MIPS) in preterm infants: preliminary data. Pediatric Research 2002;52:825.
References to studies excluded from this review
Varvarigou 1996 {published data only}
-
- Varvarigou A, Bardin CL, Beharry K, Chemtob S, Papageorgiou A, Aranda JV. Early ibuprofen administration to prevent ductus arteriosus in premature newborn infants. Journal of the American Medical Association 1996;275(7):539‐44. [PUBMED: 8606475] - PubMed
Additional references
Bell 1978
Betkerur 1981
-
- Betkerur MV, Yeh TF, Miller K, Glasser RJ, Pildes RS. Indomethacin and its effect on renal function and urinary kallikrein excretion in premature infants with patent ductus arteriosus. Pediatrics 1981;68(1):99‐102. [PUBMED: 6909683] - PubMed
Chemtob 1990
-
- Chemtob S, Beharry K, Barna T, Varma DR, Aranda JV. Prostanoids determine the range of cerebral blood flow autoregulation of newborn piglets. Stroke 1990;21(5):777‐84. [PUBMED: 2339458] - PubMed
Chemtob 1993
-
- Chemtob S, Roy MS, Abran D, Fernandez H, Varma DR. Prevention of post asphyxial increase in lipid peroxides and retinal function and deterioration in the newborn pig by inhibition of cyclooxygenase activity and free radical generation. Pediatric Research 1993;33(4 Pt 1):336‐40. [DOI: 10.1203/00006450-199304000-00006; PUBMED: 8479812] - DOI - PubMed
Clyman 2000
Coceani 1979
-
- Coceani F, White E, Bodach E, Olley PM. Age dependent changes in the response of lamb ductus arteriosus to oxygen and ibuprofen. Canadian Journal of Physiology and Pharmacology 1979;57(8):825‐31. [PUBMED: 497895] - PubMed
Coceani 2005
Cotton 1979
-
- Cotton RB, Stahlman MT, Kovar I, Catterton WZ. Medical management of small preterm infants with symptomatic patent ductus arteriosus. Journal of Pediatrics 1978;2(3):467‐73. [PUBMED: 632994] - PubMed
Couser 1996
-
- Couser RJ, Ferrara TB, Wright GB, Cabalka AK, Schilling CG, Hoekstra RE, et al. Prophylactic Indomethacin therapy in the first twenty four hours of life for the prevention of patent ductus arteriosus in preterm infants treated prophylactically with surfactant in the delivery room. Journal of Pediatrics 1996;128(5 Pt 1):631‐7. [PUBMED: 8627434] - PubMed
Davis 1995
-
- Davis P, Turner‐Gomes S, Cunningham K, Way C, Roberts R, Schmidt B. Precision and accuracy of clinical and radiological signs in premature infants at risk of patent ductus arteriosus. Archives of Pediatric and Adolescent Medicine 1995;149(10):1136‐41. [PUBMED: 7550818] - PubMed
Di Micco 2018
Domanico 1994
-
- Domanico RS, Waldman JD, Lester LA, McPhillips HA, Catrambone JE, Covert RF. Prophylactic indomethacin reduces the incidence of pulmonary hemorrhage and patent ductus arteriosus in surfactant treated < 1250g. Pediatric Research 1994;35:331A.
Edwards 1990
-
- Edwards AD, Wyatt JS, Richardson C, Potter A, Cope M, Delply DT, et al. Effects of indomethacin on cerebral hemodynamics in very preterm infants. Lancet 1990;335(8704):1491‐5. [PUBMED: 1972434] - PubMed
Fowlie 2010
Friedman 1976
Gersony 1983
-
- Gersony WM, Peckham GJ, Ellison RC, Miettinen OS, Nadas AS. Effects of Indomethacin in premature infants with patent ductus arteriosus: results of a national collaborative study. Journal of Pediatrics 1983;102(6):895‐906. [PUBMED: 6343572] - PubMed
Grosfeld 1996
Hammerman 1995
-
- Hammerman C. Patent ductus arteriosus: clinical relevance of prostaglandins and prostaglandin inhibitors in PDA pathophysiology and treatment. Clinics in Perinatology 1995;22(2):457‐79. [PUBMED: 7671547] - PubMed
Higgins 2003
Higgins 2017
-
- Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.2.0 (updated June 2017). The Cochrane Collaboration, 2017. Available from training.cochrane.org/handbook.
ICROP 1984
-
- Committee for the Classification of Retinopathy of Prematurity. An international classification of retinopathy of prematurity. Archives of Ophthalmology 1984;102(8):1130‐4. [PUBMED: 6547831] - PubMed
Ito 1994
-
- Ito K, Niida Y, Sato J, Owada E, Ito K, Umetsu M. Pharmacokinetics of mefenamic acid in preterm infants with patent ductus arteriosus. Acta Paediatrica Japonica 1994;36(4):387‐91. [PUBMED: 7942001] - PubMed
Jones 2011
Lago 2002
-
- Lago P, Bettiol T, Salvadori S, Pitassi I, Vianello A, Chiandetti L, et al. Safety and efficacy of ibuprofen versus indomethacin in preterm infants treated for patent ductus arteriosus: a randomised controlled trial. European Journal of Pediatrics 2002;161(4):202‐7. [PUBMED: 12014386] - PubMed
Lee 2000
-
- Lee Sk, McMillan DD, Ohlsson A, Pendray M, Synnes A, Whyte R, et al. Variations in practice and outcomes in the Canadian NICU network: 1996‐1997. Pediatrics 2000;106(5):1070‐9. [PUBMED: 11061777] - PubMed
Mosca 1997
-
- Mosca F, Bray M, Lattnazio M, Fumagalli M, Toscetto C. Comparative evaluation of the effect of indomethacin and ibuprofen on cerebral perfusion and oxygenation in preterm infants with patent ductus arteriosus. Journal of Pediatrics 1997;131(4):549‐54. [PUBMED: 9386657] - PubMed
Mosca 2002
-
- Mosca F, Bray M, Stucchi I, Fumagalli M. Pulmonary hypertension after ibuprofen prophylaxis in very preterm infants. Lancet 2002;9338:1023‐4. - PubMed
Niopas 1994
-
- Niopas I, Mamzoridi K. Determination of indomethacin and mefenamic acid in plasma performance liquid chromatography. Journal of Chromatography. Biomedical Applications 1994;656(2):447‐50. [PUBMED: 7987501] - PubMed
Ohlsson 1993
-
- Ohlsson A, Bottu J, Govan J, Ryan ML, Fong K, Myhr T. The effect of indomethacin on cerebral blood flow velocities in very low birth weight neonates with patent ductus arteriosus. Developmental Pharmacology and Therapeutics 1993;20(1‐2):100‐6. [PUBMED: 7924757] - PubMed
Ohlsson 2018
Pai 2008
Papile 1978
-
- Papile LA, Burstein J, Burstein R, Koffler H. Incidence and evolution of subependymal and intraventricular hemorrhage: a study of infants with birthweights less than 1,500 grams. Journal of Pediatrics 1978;92(4):529‐34. [PUBMED: 305471] - PubMed
Patel 2000
-
- Patel J, Roberts I, Azzopardi D, Hamilton P, Edwards AD. Randomised double blind controlled trial comparing the effects of ibuprofen with indomethacin on cerebral hemodynamics in preterm infants with patent ductus arteriosus. Pediatric Research 2000;47(1):36‐42. [PUBMED: 10625080] - PubMed
Pezzati 1999
-
- Pezzati M, Vangi V, Biagiotti R, Bertini G, Cianciulli D, Rubaltelli FF. Effects of indomethacin and ibuprofen on mesenteric and renal blood flow in preterm infants with patent ductus arteriosus. Journal of Pediatrics 1999;135(6):733‐8. [PUBMED: 10586177] - PubMed
Ramanathan 1997
-
- Ramanathan R, Siassi B, Gallagher R, DeLemos RA. Outcome of very low birth weight infants < 1500 g enrolled in the national database network: are there any trends in neonatology?. Pediatric Research 1997;41:171A.
Sakhalkar 1992
-
- Sakhalkar VS, Merchant RH. Therapy of patent ductus arteriosus in preterms with mefenamic acid and indomethacin. Indian Pediatrics 1992;29(3):313‐8. [PUBMED: 1612672] - PubMed
Schmidt 2001
Schünemann 2013
-
- Schünemann H, Brożek J, Guyatt G, Oxman A, editor(s). Handbook for grading the quality of evidence and the strength of recommendations using the GRADE approach (updated October 2013). GRADE Working Group, 2013. Available from gdt.guidelinedevelopment.org/app/handbook/handbook.html.
Shennan 1988
-
- Shennan AT, Dunn MS, Ohlsson A, Lennox K, Hoskins EM. Abnormal pulmonary outcomes in premature infants: prediction from oxygen requirement in the neonatal period. Pediatrics 1988;82(4):527‐32. [PUBMED: 3174313] - PubMed
Speziale 1999
Van Bel 1989
-
- Bel F, Bor M, Stijnen T, Baan J, Ruys JH. Cerebral blood flow velocity changes in preterm infants after a single dose of indomethacin: duration of its effect. Pediatrics 1989;84(5):802‐7. [PUBMED: 2677960] - PubMed
Van Overmeire 1997
Van Overmeire 2000
-
- Overmeire B, Smets K, Lecoutere D, Broek H, Weyler J, Groote K, et al. A comparison of ibuprofen and indomethacin for closure of patent ductus arteriosus. New England Journal of Medicine 2000;334:674‐81. - PubMed
References to other published versions of this review
Ohlsson 2009
Ohlsson 2011
Review Manager 5 (RevMan 5) [Computer program]
-
- Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager 5 (RevMan 5). Version 5.3. Copenhagen: Nordic Cochrane Centre, The Cochrane Collaboration, 2014.
Shah 2003
Shah 2007
-
- Shah SS, Ohlsson A. Ibuprofen for the prevention of patent ductus arteriosus in preterm and/or low birth weight infants. Cochrane Database of Systematic Reviews 2006, Issue 1. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

