Validation of the Data Collection on Adverse Events of Anti-HIV Drugs (D:A:D) chronic kidney disease risk score in HIV-infected patients in the USA
- PMID: 31985887
- PMCID: PMC7217174
- DOI: 10.1111/hiv.12826
Validation of the Data Collection on Adverse Events of Anti-HIV Drugs (D:A:D) chronic kidney disease risk score in HIV-infected patients in the USA
Abstract
Objectives: The aim of the study was to assess the validity of an easy-to-calculate chronic kidney disease (CKD) risk score developed by the Data Collection on Adverse Events of Anti-HIV Drugs (D:A:D) group in a longitudinal observational study of people living with HIV (PLWH) in the USA.
Methods: PLWH (2002-2016) without prior exposure to potentially nephrotoxic antiretroviral agents and with at least three estimated glomerular filtration rate (eGFR) test results were identified in the Observational Pharmaco-Epidemiology Research and Analysis (OPERA® ) cohort. Three samples were drawn independently using the same eligibility criteria but each using a different eGFR equation, specifically the Cockcroft-Gault (C-G), Modification of Diet in Renal Disease (MDRD) or Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) eGFR estimation method. Full and short D:A:D risk scores were applied. CKD was defined as a confirmed decrease in eGFR to < 60 mL/min/1.73 m2 (stages 3-5). Poisson models estimated the association between CKD incidence and a one-point increase in the continuous risk score. The incidence rate ratio (IRR), adjusted IRR (aIRR), and Harrell's discrimination statistic were used to assess validity.
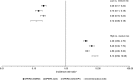
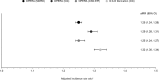
Results: There were 19 444, 22 727 and 22 748 PLWH in the OPERA C-G, CKD-EPI and MDRD samples, respectively. The median (minimum-maximum) follow-up duration was 6.1 (0.3-9.1) years in the D:A:D cohort and ranged from 3.2 to 3.5 (0.2-15.5) years in the OPERA validation samples. The observation time for the majority of PLWH in the D:A:D cohort began prior to 2006, in stark contrast to the OPERA validation samples, where the majority of PLWH were observed after 2011. The CKD incidence ranged from 7.3 per 1000 person-years [95% confidence interval (CI) 6.8, 7.9 per 1000 person-years] in OPERA C-G to 11.0 (95% CI 10.4, 11.6 per 1000 person-years) in OPERA MDRD. In OPERA samples, IRRs by risk group and adjusted IRRs (full risk score) were similar to those in the D:A:D derivation cohort (adjusted IRR 1.3; 95% CI 1.3, 1.3). Harrell's c-statistic ranged from 0.87 to 0.92 in the OPERA samples, comparable to that in the derivation cohort (0.92). Results for short scores were similar.
Conclusions: The findings support the validity of the D:A:D risk scoring method for assessing CKD (stages 3-5) probability in an exclusively USA-based sample regardless of eGFR method.
Keywords: Data Collection on Adverse Events of Anti-HIV Drugs (D:A:D) risk score; HIV; chronic kidney disease; renal impairment; validation.
© 2020 The Authors. HIV Medicine published by John Wiley & Sons Ltd on behalf of British HIV Association.
Figures


Similar articles
-
Risk of chronic kidney disease in people living with HIV by tenofovir disoproxil fumarate (TDF) use and baseline D:A:D chronic kidney disease risk score.HIV Med. 2021 May;22(5):325-333. doi: 10.1111/hiv.13019. Epub 2020 Nov 28. HIV Med. 2021. PMID: 33247876 Free PMC article.
-
Development and validation of a risk score for chronic kidney disease in HIV infection using prospective cohort data from the D:A:D study.PLoS Med. 2015 Mar 31;12(3):e1001809. doi: 10.1371/journal.pmed.1001809. eCollection 2015 Mar. PLoS Med. 2015. PMID: 25826420 Free PMC article.
-
Chronic kidney disease and HIV in the era of antiretroviral treatment: findings from a 10-year cohort study in a west African setting.BMC Nephrol. 2019 May 7;20(1):155. doi: 10.1186/s12882-019-1335-9. BMC Nephrol. 2019. PMID: 31064340 Free PMC article.
-
Chronic Kidney Disease and Antiretroviral Therapy in HIV-Positive Individuals: Recent Developments.Curr HIV/AIDS Rep. 2016 Jun;13(3):149-57. doi: 10.1007/s11904-016-0315-y. Curr HIV/AIDS Rep. 2016. PMID: 27130284 Review.
-
Contemporary issues and new challenges in chronic kidney disease amongst people living with HIV.AIDS Res Ther. 2020 Mar 16;17(1):11. doi: 10.1186/s12981-020-00266-3. AIDS Res Ther. 2020. PMID: 32178687 Free PMC article. Review.
Cited by
-
Incidence of impaired kidney function among people with HIV: a systematic review and meta-analysis.BMC Nephrol. 2022 Mar 17;23(1):107. doi: 10.1186/s12882-022-02721-x. BMC Nephrol. 2022. PMID: 35300612 Free PMC article.
-
Risk of chronic kidney disease in people living with HIV by tenofovir disoproxil fumarate (TDF) use and baseline D:A:D chronic kidney disease risk score.HIV Med. 2021 May;22(5):325-333. doi: 10.1111/hiv.13019. Epub 2020 Nov 28. HIV Med. 2021. PMID: 33247876 Free PMC article.
-
Validation of the D:A:D chronic kidney disease risk score in people living with HIV: the IeDEA West Africa Cohort Collaboration.HIV Med. 2021 Feb;22(2):113-121. doi: 10.1111/hiv.12982. Epub 2020 Nov 3. HIV Med. 2021. PMID: 33145918 Free PMC article.
-
Validation of the D: A: D Chronic Kidney Disease Risk Score Model Among People Living With HIV in the Asia-Pacific.J Acquir Immune Defic Syndr. 2020 Dec 1;85(4):489-497. doi: 10.1097/QAI.0000000000002464. J Acquir Immune Defic Syndr. 2020. PMID: 33136750 Free PMC article.
References
-
- Kooij KW, Vogt L, Wit F et al Higher prevalence and faster progression of chronic kidney disease in human immunodeficiency virus‐infected middle‐aged individuals compared with human immunodeficiency virus‐uninfected controls. J Infect Dis 2017; 216 (6): 622–631. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

