Fetal and amniotic fluid iron homeostasis in healthy and complicated murine, macaque, and human pregnancy
- PMID: 31990688
- PMCID: PMC7101151
- DOI: 10.1172/jci.insight.135321
Fetal and amniotic fluid iron homeostasis in healthy and complicated murine, macaque, and human pregnancy
Abstract
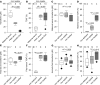
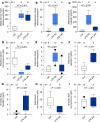
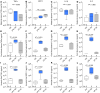
Adequate iron supply during pregnancy is essential for fetal development. However, how fetal or amniotic fluid iron levels are regulated during healthy pregnancy, or pregnancies complicated by intraamniotic infection or inflammation (IAI), is unknown. We evaluated amniotic fluid and fetal iron homeostasis in normal and complicated murine, macaque, and human pregnancy. In mice, fetal iron endowment was affected by maternal iron status, but amniotic fluid iron concentrations changed little during maternal iron deficiency or excess. In murine and macaque models of inflamed pregnancy, the fetus responded to maternal systemic inflammation or IAI by rapidly upregulating hepcidin and lowering iron in fetal blood, without altering amniotic fluid iron. In humans, elevated cord blood hepcidin with accompanying hypoferremia was observed in pregnancies with antenatal exposure to IAI compared with those that were nonexposed. Hepcidin was also elevated in human amniotic fluid from pregnancies with IAI compared with those without IAI, but amniotic fluid iron levels did not differ between the groups. Our studies in mice, macaques, and humans demonstrate that amniotic fluid iron is largely unregulated but that the rapid induction of fetal hepcidin by inflammation and consequent fetal hypoferremia are conserved mechanisms that may be important in fetal host defense.
Keywords: Bacterial infections; Homeostasis; Innate immunity; Reproductive Biology.
Conflict of interest statement
Figures



References
-
- Legge M. Second trimester amniotic fluid ferritin and transferrin levels in normal and anencephalic pregnancies. J Obstet Gynaecol (Lahore) 1983;4(1):14–15. doi: 10.3109/01443618309071216. - DOI

