The Role of Lozenge in Drosophila Hematopoiesis
- PMID: 31992020
- PMCID: PMC7057836
- DOI: 10.14348/molcells.2019.0249
The Role of Lozenge in Drosophila Hematopoiesis
Abstract
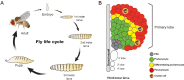
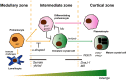
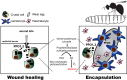
Drosophila hematopoiesis is comparable to mammalian differentiation of myeloid lineages, and therefore, has been a useful model organism in illustrating the molecular and genetic basis for hematopoiesis. Multiple novel regulators and signals have been uncovered using the tools of Drosophila genetics. A Runt domain protein, lozenge, is one of the first players recognized and closely studied in the hematopoietic lineage specification. Here, we explore the role of lozenge in determination of prohemocytes into a special class of hemocyte, namely the crystal cell, and discuss molecules and signals controlling the lozenge function and its implication in immunity and stress response. Given the highly conserved nature of Runt domain in both invertebrates and vertebrates, studies in Drosophila will enlighten our perspectives on Runx-mediated development and pathologies.
Keywords: Drosophila melanogaster; RUNX; crystal cells; hematopoiesis; lozenge; lymph gland; melanization; prophenoloxidase.
Conflict of interest statement
The authors have no potential conflicts of interest to disclose.
Figures
Similar articles
-
Combinatorial interactions of serpent, lozenge, and U-shaped regulate crystal cell lineage commitment during Drosophila hematopoiesis.Proc Natl Acad Sci U S A. 2003 Sep 30;100(20):11451-6. doi: 10.1073/pnas.1635050100. Epub 2003 Sep 22. Proc Natl Acad Sci U S A. 2003. PMID: 14504400 Free PMC article.
-
Regulation of Drosophila friend of GATA gene, u-shaped, during hematopoiesis: a direct role for serpent and lozenge.Dev Biol. 2006 Aug 15;296(2):561-79. doi: 10.1016/j.ydbio.2006.04.455. Epub 2006 Apr 27. Dev Biol. 2006. PMID: 16730345
-
The GATA factor Serpent cross-regulates lozenge and u-shaped expression during Drosophila blood cell development.Dev Biol. 2007 Nov 15;311(2):636-49. doi: 10.1016/j.ydbio.2007.08.015. Epub 2007 Aug 16. Dev Biol. 2007. PMID: 17869239 Free PMC article.
-
Runt and Lozenge function in Drosophila development.Semin Cell Dev Biol. 2000 Oct;11(5):327-36. doi: 10.1006/scdb.2000.0185. Semin Cell Dev Biol. 2000. PMID: 11105896 Review.
-
Thicker than blood: conserved mechanisms in Drosophila and vertebrate hematopoiesis.Dev Cell. 2003 Nov;5(5):673-90. doi: 10.1016/s1534-5807(03)00335-6. Dev Cell. 2003. PMID: 14602069 Review.
Cited by
-
Mechanism of hyperproteinemia-induced blood cell homeostasis imbalance in an animal model.Zool Res. 2022 May 18;43(3):301-318. doi: 10.24272/j.issn.2095-8137.2021.397. Zool Res. 2022. PMID: 35312240 Free PMC article.
-
Transcription factor expression landscape in Drosophila embryonic cell lines.BMC Genomics. 2024 Mar 23;25(1):307. doi: 10.1186/s12864-024-10241-1. BMC Genomics. 2024. PMID: 38521929 Free PMC article.
-
Hemocyte Clusters Defined by scRNA-Seq in Bombyx mori: In Silico Analysis of Predicted Marker Genes and Implications for Potential Functional Roles.Front Immunol. 2022 Feb 25;13:852702. doi: 10.3389/fimmu.2022.852702. eCollection 2022. Front Immunol. 2022. PMID: 35281044 Free PMC article.
-
Single-cell analysis of mosquito hemocytes identifies signatures of immune cell subtypes and cell differentiation.Elife. 2021 Jul 28;10:e66192. doi: 10.7554/eLife.66192. Elife. 2021. PMID: 34318744 Free PMC article.
-
The Roles of the LIM Domain Proteins in Drosophila Cardiac and Hematopoietic Morphogenesis.Front Cardiovasc Med. 2021 Feb 11;8:616851. doi: 10.3389/fcvm.2021.616851. eCollection 2021. Front Cardiovasc Med. 2021. PMID: 33681304 Free PMC article. Review.
References
-
- Ashburner M., Novitski E. Academic Press; London: 1976. Genetics and Biology of Drosophila.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

