The physiological role of Wnt pathway in normal development and cancer
- PMID: 31996036
- PMCID: PMC7082880
- DOI: 10.1177/1535370220901683
The physiological role of Wnt pathway in normal development and cancer
Abstract
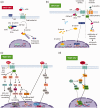
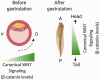
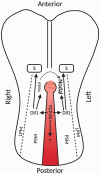
Over the decades, many studies have illustrated the critical roles of Wnt signaling pathways in both developmental processes as well as tumorigenesis. Due to the complexity of Wnt signaling regulation, there are still questions to be addressed about ways cells are able to manipulate different types of Wnt pathways in order to fulfill the requirements for normal or cancer development. In this review, we will describe different types of Wnt signaling pathways and their roles in both normal developmental processes and their role in cancer development and progression. Additionally, we will briefly introduce new strategies currently in clinical trials targeting Wnt signaling pathway components for cancer therapy.
Impact statement: Wnt pathway does not only play a critical role in mammalian development but has been hijacked by cancer cells and the tumor microenvironment to promote tumor progression and metastasis. Recent evidence supports further interrogation of the Wnt pathway for bench-to-bedside translation into cancer therapy. This review highlights the role of the Wnt pathway in normal development and tumorigenesis, along with an overview of new therapies currently undergoing clinical trials.
Keywords: Wnt signaling; development; tumorigenesis.
Figures


References
-
- Sharma RP, Chopra VL. Effect of the wingless (wg1) mutation on wing and haltere development in Drosophila melanogaster. Dev Biol 1976; 48:461–5 - PubMed
-
- Nusslein-Volhard C, Wieschaus E. Mutations affecting segment number and polarity in Drosophila. Nature 1980; 287:795–801 - PubMed
-
- Nusse R, Varmus HE. Many tumors induced by the mouse mammary tumor virus contain a provirus integrated in the same region of the host genome. Cell 1982; 31:99–109 - PubMed
-
- Rijsewijk F, Schuermann M, Wagenaar E, Parren P, Weigel D, Nusse R. The Drosophila homolog of the mouse mammary oncogene int-1 is identical to the segment polarity gene wingless. Cell 1987; 50:649–57 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

