Bolus norepinephrine and phenylephrine for maternal hypotension during elective cesarean section with spinal anesthesia: a randomized, double-blinded study
- PMID: 31996543
- PMCID: PMC7065858
- DOI: 10.1097/CM9.0000000000000621
Bolus norepinephrine and phenylephrine for maternal hypotension during elective cesarean section with spinal anesthesia: a randomized, double-blinded study
Abstract
Background: In recent years, norepinephrine has attracted increasing attention for the management of maternal hypotension during elective cesarean section with spinal anesthesia. Intermittent bolus is a widely used administration paradigm for vasopressors in obstetric anesthesia in China. Thus, in this randomized, double-blinded study, we compared the efficacy and safety of equivalent bolus norepinephrine and phenylephrine for rescuing maternal post-spinal hypotension.
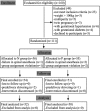
Methods: In a tertiary women's hospital in Nanjing, China, 102 women were allocated with computer derived randomized number to receive prophylactic 8 μg norepinephrine (group N; n = 52) or 100 μg phenylephrine (group P; n = 50) immediately post-spinal anesthesia, followed by an extra bolus of the same dosage until delivery whenever maternal systolic blood pressure became lower than 80% of the baseline. Our primary outcome was standardized maternal cardiac output (CO) reading from spinal anesthesia until delivery analyzed by a two-step method. Other hemodynamic parameters related to vasopressor efficacy and safety were considered as secondary outcomes. Maternal side effects and neonatal outcomes were collected as well.
Results: Compared to group P, women in group N had a higher CO (standardized CO 5.8 ± 0.9 vs. 5.3 ± 1.0 L/min, t = 2.37, P = 0.02) and stroke volume (SV, standardized SV 73.6 ± 17.2 vs. 60.0 ± 13.3 mL, t = 4.52, P < 0.001), and a lower total peripheral resistance (875 ± 174 vs. 996 ± 182 dyne·s/cm, t = 3.44, P < 0.001). Furthermore, the incidence of bradycardia was lower in group N than in group P (2% vs. 14%, P = 0.023), along with an overall higher standardized heart rate (78.8 ± 11.6 vs. 75.0 ± 7.3 beats/min, P = 0.049). Other hemodynamics, as well as maternal side effects and neonatal outcomes, were similar in two groups (P > 0.05).
Conclusions: Compared to equivalent phenylephrine, intermittent bolus norepinephrine provides a greater CO for management of maternal hypotension during elective cesarean section with spinal anesthesia; however, no obvious maternal or neonatal clinical advantages were observed for norepinephrine.
Conflict of interest statement
None.
Figures


Similar articles
-
A Comparative Study of Bolus Norepinephrine, Phenylephrine, and Ephedrine for the Treatment of Maternal Hypotension in Parturients with Preeclampsia During Cesarean Delivery Under Spinal Anesthesia.Med Sci Monit. 2019 Feb 9;25:1093-1101. doi: 10.12659/MSM.914143. Med Sci Monit. 2019. PMID: 30738019 Free PMC article.
-
An open-label randomized controlled clinical trial for comparison of continuous phenylephrine versus norepinephrine infusion in prevention of spinal hypotension during cesarean delivery.Int J Obstet Anesth. 2017 Feb;29:18-25. doi: 10.1016/j.ijoa.2016.08.005. Epub 2016 Aug 28. Int J Obstet Anesth. 2017. PMID: 27720613 Clinical Trial.
-
Norepinephrine versus phenylephrine infusion for prophylaxis against post-spinal anaesthesia hypotension during elective caesarean delivery: A randomised controlled trial.Anaesth Crit Care Pain Med. 2019 Dec;38(6):601-607. doi: 10.1016/j.accpm.2019.03.005. Epub 2019 Mar 30. Anaesth Crit Care Pain Med. 2019. PMID: 30935897 Clinical Trial.
-
The Efficacy and Safety of Norepinephrine and Its Feasibility as a Replacement for Phenylephrine to Manage Maternal Hypotension during Elective Cesarean Delivery under Spinal Anesthesia.Biomed Res Int. 2018 Dec 31;2018:1869189. doi: 10.1155/2018/1869189. eCollection 2018. Biomed Res Int. 2018. PMID: 30687737 Free PMC article. Review.
-
Efficacy and safety of norepinephrine versus phenylephrine for the management of maternal hypotension during cesarean delivery with spinal anesthesia: A systematic review and meta-analysis.Medicine (Baltimore). 2019 Feb;98(5):e14331. doi: 10.1097/MD.0000000000014331. Medicine (Baltimore). 2019. PMID: 30702617 Free PMC article.
Cited by
-
Comparison of phenylephrine, ephedrine, and norepinephrine for the prevention and treatment of spinal-induced hypotension in pre-eclamptic patients undergoing caesarean section: A systematic review and network meta-analysis.Indian J Anaesth. 2025 Jun;69(6):526-539. doi: 10.4103/ija.ija_62_25. Epub 2025 May 14. Indian J Anaesth. 2025. PMID: 40470387 Free PMC article.
-
Comparison of phenylephrine and norepinephrine for treatment of spinal hypotension during elective cesarean delivery- A randomised, double-blind study.J Anaesthesiol Clin Pharmacol. 2022 Jul-Sep;38(3):445-452. doi: 10.4103/joacp.JOACP_499_20. Epub 2022 Feb 10. J Anaesthesiol Clin Pharmacol. 2022. PMID: 36505203 Free PMC article.
-
Up-and-down determination of prophylactic norepinephrine boluses combined with crystalloid co-load for preventing post-spinal anesthesia hypotension during cesarean section.BMC Anesthesiol. 2025 Jan 25;25(1):38. doi: 10.1186/s12871-025-02913-6. BMC Anesthesiol. 2025. PMID: 39856541 Free PMC article. Clinical Trial.
-
Comparison of Prophylactic Norepinephrine and Phenylephrine Infusions During Spinal Anaesthesia for Primary Caesarean Delivery in Twin Pregnancies: A Randomized Double-Blinded Clinical Study.Drug Des Devel Ther. 2022 Mar 24;16:789-798. doi: 10.2147/DDDT.S357507. eCollection 2022. Drug Des Devel Ther. 2022. PMID: 35355656 Free PMC article. Clinical Trial.
-
A Comparison of Norepinephrine versus Phenylephrine to Prevent Hypotension after Spinal Anesthesia for Cesarean Section: Systematic Review and Meta-Analysis.J Pers Med. 2024 Jul 29;14(8):803. doi: 10.3390/jpm14080803. J Pers Med. 2024. PMID: 39201995 Free PMC article. Review.
References
-
- Macarthur A, Riley E. Obstetric anesthesia controversies: vasopressor choice for postspinal hypotension during cesarean delivery. Int Anesthesiol Clin 2007; 45:115–132. doi: 10.1097/AIA.0b013e31802b8d53. - PubMed
-
- Xu S, Shen X, Liu S, Yang J, Wang X. Efficacy and safety of norepinephrine versus phenylephrine for the management of maternal hypotension during cesarean delivery with spinal anesthesia: a systematic review and meta-analysis. Medicine 2019; 98:e14331.doi: 10.1097/MD.0000000000014331. - PMC - PubMed
-
- Kinsella SM, Carvalho B, Dyer RA, Fernando R, Mcdonnell N, Mercier FJ, et al. International consensus statement on the management of hypotension with vasopressors during caesarean section under spinal anaesthesia. Anaesthesia 2018; 73:71–92. doi: 10.1111/anae.14080. - PubMed
-
- Allen TK, Muir HA, George RB, Habib AS. A survey of the management of spinal-induced hypotension for scheduled cesarean delivery. Int J Obstet Anesth 2009; 18:356–361. doi: 10.1016/j.ijoa.2009.03.014. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

