Presynaptic Boutons That Contain Mitochondria Are More Stable
- PMID: 31998110
- PMCID: PMC6966497
- DOI: 10.3389/fnsyn.2019.00037
Presynaptic Boutons That Contain Mitochondria Are More Stable
Abstract
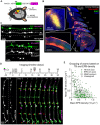
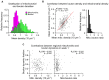
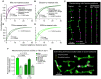
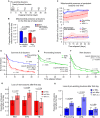
The addition and removal of presynaptic terminals reconfigures neuronal circuits of the mammalian neocortex, but little is known about how this presynaptic structural plasticity is controlled. Since mitochondria can regulate presynaptic function, we investigated whether the presence of axonal mitochondria relates to the structural plasticity of presynaptic boutons in mouse neocortex. We found that the overall density of axonal mitochondria did not appear to influence the loss and gain of boutons. However, positioning of mitochondria at individual presynaptic sites did relate to increased stability of those boutons. In line with this, synaptic localization of mitochondria increased as boutons aged and showed differing patterns of localization at en passant and terminaux boutons. These results suggest that mitochondria accumulate locally at boutons over time to increase bouton stability.
Keywords: in vivo 2-photon imaging; mitochondria; neocortex; presynaptic bouton; synapse turnover; synaptic plasticity.
Copyright © 2020 Lees, Johnson and Ashby.
Figures
Similar articles
-
Long-term stability of axonal boutons in the mouse barrel cortex.Dev Neurobiol. 2016 Mar;76(3):252-61. doi: 10.1002/dneu.22311. Epub 2015 Jun 12. Dev Neurobiol. 2016. PMID: 26058471 Free PMC article.
-
Cell type-specific structural plasticity of axonal branches and boutons in the adult neocortex.Neuron. 2006 Mar 16;49(6):861-75. doi: 10.1016/j.neuron.2006.02.017. Neuron. 2006. PMID: 16543134
-
Cortical Presynaptic Boutons Progressively Engulf Spinules as They Mature.eNeuro. 2020 Oct 15;7(5):ENEURO.0426-19.2020. doi: 10.1523/ENEURO.0426-19.2020. Print 2020 Sep/Oct. eNeuro. 2020. PMID: 32958478 Free PMC article.
-
[Recent advances of strategy for the brain functions--a targeting for tiny presynaptic boutons].Nihon Yakurigaku Zasshi. 2002 Nov;120(1):16P-19P. Nihon Yakurigaku Zasshi. 2002. PMID: 12491769 Review. Japanese.
-
Regulation of axonal mitochondrial transport and its impact on synaptic transmission.Neurosci Res. 2011 May;70(1):9-15. doi: 10.1016/j.neures.2011.02.005. Epub 2011 Feb 23. Neurosci Res. 2011. PMID: 21352858 Free PMC article. Review.
Cited by
-
Mitochondrial support and local translation of mitochondrial proteins in synaptic plasticity and function.Histol Histopathol. 2021 Oct;36(10):1007-1019. doi: 10.14670/HH-18-345. Epub 2021 May 25. Histol Histopathol. 2021. PMID: 34032272 Review.
-
Mitochondrial heterogeneity and homeostasis through the lens of a neuron.Nat Metab. 2022 Jul;4(7):802-812. doi: 10.1038/s42255-022-00594-w. Epub 2022 Jul 11. Nat Metab. 2022. PMID: 35817853 Free PMC article. Review.
-
Presynaptic stochasticity improves energy efficiency and helps alleviate the stability-plasticity dilemma.Elife. 2021 Oct 18;10:e69884. doi: 10.7554/eLife.69884. Elife. 2021. PMID: 34661525 Free PMC article.
-
Calcium signaling in postsynaptic mitochondria: mechanisms, dynamics, and role in ATP production.Front Mol Neurosci. 2025 Jul 21;18:1621070. doi: 10.3389/fnmol.2025.1621070. eCollection 2025. Front Mol Neurosci. 2025. PMID: 40761376 Free PMC article. Review.
-
Specializations in Amygdalar and Hippocampal Innervation of the Primate Nucleus Accumbens Shell.J Neurosci. 2025 Jun 11;45(24):e2425242025. doi: 10.1523/JNEUROSCI.2425-24.2025. J Neurosci. 2025. PMID: 40374561
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

