The tumour response of postmenopausal hormone receptor-positive breast cancers undergoing different types of neoadjuvant therapy: a meta-analysis
- PMID: 32005117
- PMCID: PMC6993383
- DOI: 10.1186/s12905-020-0879-y
The tumour response of postmenopausal hormone receptor-positive breast cancers undergoing different types of neoadjuvant therapy: a meta-analysis
Abstract
Background: To investigate the efficacy of neoadjuvant chemotherapy (NCT), neoadjuvant endocrine therapy (NET) and neoadjuvant chemoendocrine therapy (NCET) on the tumour response, including pathological complete response (pCR) rate and overall response rate (ORR), in postmenopausal women with hormone receptor (HR)-positive breast cancer.
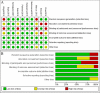
Methods: Based on a PRISMA-IPD statement, the PubMed, Embase and Cochrane Library databases were used to identify eligible trials published from inception to 7 May 2019. Pooled odds ratio (OR) with 95% confidential interval (CI) was calculated to assess the pCR rate and ORR of tumours among those three treatments via fixed- or random-effect Mantel-Haenszel models in terms of a Heterogeneity Chi2 test with a significant level of p < 0.1. All statistical tests were performed by the software of StataSE, version 12.0.
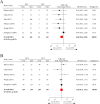
Results: The analysed data consisted of 10 eligible clinical trials with 971 unique HR-positive breast cancer patients. The pooled results indicated that the pCR rate of those patients undergoing NET was significantly lower than those undergoing NCT (pooled OR, 0.48; 95% CI, 0.26-0.90), whereas the difference of ORR between both therapies was not statistically significant (pooled OR, 1.05; 95% CI, 0.73-1.52). The combined paradigm of NCET compared with the monotherapy of NET or NCT did not present a significantly improved pCR rate or ORR (pooled OR, 2.61; 95% CI, 0.94-7.25; and 2.25; 95% CI, 0.39-13.05; respectively).
Conclusion: Postmenopausal HR-positive breast cancer patients after NCT may have better tumour response than those after NET, while those undergoing NCET may not manifest the apparently improved clinical efficacies compared to those receiving monotherapy.
Keywords: Breast cancer; Neoadjuvant chemoendocrine therapy; Neoadjuvant chemotherapy; Neoadjuvant endocrine therapy; Tumour response.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures


References
-
- Wolmark N, Wang J, Mamounas E, Bryant J, Fisher B. Preoperative chemotherapy in patients with operable breast cancer: nine-year results from National Surgical Adjuvant Breast and bowel project B-18. J Natl Cancer Inst Monogr. 2001;30:96–102. doi: 10.1093/oxfordjournals.jncimonographs.a003469. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

