Oleic acid-based nanosystems for mitigating acute respiratory distress syndrome in mice through neutrophil suppression: how the particulate size affects therapeutic efficiency
- PMID: 32005196
- PMCID: PMC6995149
- DOI: 10.1186/s12951-020-0583-y
Oleic acid-based nanosystems for mitigating acute respiratory distress syndrome in mice through neutrophil suppression: how the particulate size affects therapeutic efficiency
Abstract
Background: Oleic acid (OA) is reported to show anti-inflammatory activity toward activated neutrophils. It is also an important material in nanoparticles for increased stability and cellular internalization. We aimed to evaluate the anti-inflammatory activity of injectable OA-based nanoparticles for treating lung injury. Different sizes of nanocarriers were prepared to explore the effect of nanoparticulate size on inflammation inhibition.
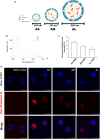
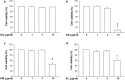
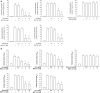
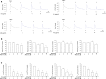
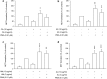
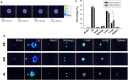
Results: The nanoparticles were fabricated with the mean diameters of 105, 153, and 225 nm. The nanocarriers were ingested by isolated human neutrophils during a 5-min period, with the smaller sizes exhibiting greater uptake. The size reduction led to the decrease of cell viability and the intracellular calcium level. The OA-loaded nanosystems dose-dependently suppressed the superoxide anion and elastase produced by the stimulated neutrophils. The inhibition level was comparable for the nanoparticles of different sizes. In the ex vivo biodistribution study, the pulmonary accumulation of nanoparticles increased following the increase of particle size. The nanocarriers were mainly excreted by the liver and bile clearance. Mice were exposed to intratracheal lipopolysaccharide (LPS) to induce acute respiratory distress syndrome (ARDS), like lung damage. The lipid-based nanocarriers mitigated myeloperoxidase (MPO) and cytokines more effectively as compared to OA solution. The larger nanoparticles displayed greater reduction on MPO, TNF-α, and IL-6 than the smaller ones. The histology confirmed the decreased pulmonary neutrophil recruitment and lung-architecture damage after intravenous administration of larger nanoparticles.
Conclusions: Nanoparticulate size, an essential property governing the anti-inflammatory effect and lung-injury therapy, had different effects on activated neutrophil inhibition and in vivo therapeutic efficacy.
Keywords: Acute respiratory distress syndrome; Anti-inflammation; Lipid-based nanoparticles; Neutrophil; Oleic acid; Size.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures

References
-
- Carrillo C, Cavia DM, Alonso-Torre S. Role of oleic acid in immune system; mechanism of action; a review. Nutr Hosp. 2012;27(4):978–990. - PubMed
-
- Gonçalves-de-Albuquerque CF, Medeiros-de-Moraes IM, de Jesus Oliveira FM, Burth P, Bozza PT, Faria MVC, Silva AR, de Castro-Faria-Neto HC. Omega-9 oleic acid induces fatty acid oxidation and decreases organ dysfunction and mortality in experimental sepsis. PLoS ONE. 2016;11(4):e0153607. doi: 10.1371/journal.pone.0153607. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

