Modification of Ammonia Decomposition Activity of Ruthenium Nanoparticles by N-Doping of CNT Supports
- PMID: 32009773
- PMCID: PMC6961473
- DOI: 10.1007/s11244-017-0806-0
Modification of Ammonia Decomposition Activity of Ruthenium Nanoparticles by N-Doping of CNT Supports
Abstract
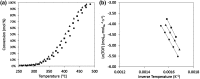
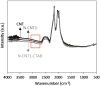
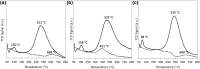
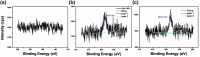
The use of ammonia as a hydrogen vector has the potential to unlock the hydrogen economy. In this context, this paper presents novel insights into improving the ammonia decomposition activity of ruthenium nanoparticles supported on carbon nanotubes (CNT) by nitrogen doping. Our results can be applied to develop more active systems capable of delivering hydrogen on demand, with a view to move towards the low temperature target of less than 150 °C. Herein we demonstrate that nitrogen doping of the CNT support enhances the activity of ruthenium nanoparticles for the low temperature ammonia decomposition with turnover frequency numbers at 400 °C of 6200 LH2 molRu -1 h-1, higher than the corresponding value of unmodified CNT supports under the same conditions (4400 LH2 molRu -1 h- 1), despite presenting similar ruthenium particle sizes. However, when the nitrogen doping process is carried out with cetyltrimethylammonium bromide (CTAB) to enhance the dispersion of CNTs, the catalyst becomes virtually inactive despite the small ruthenium particle size, likely due to interference of CTAB, weakening the metal-support interaction. Our results demonstrate that the low temperature ammonia decomposition activity of ruthenium can be enhanced by nitrogen doping of the CNT support due to simultaneously increasing the support's conductivity and basicity, electronically modifying the ruthenium active sites and promoting a strong metal-support interaction.
Keywords: Ammonia decomposition; In situ H2 production; N-CNT; Nitrogen; Nitrogen-doped CNT; Ruthenium.
© The Author(s) 2017.
Figures





References
-
- Durbin DJ, Malardier-Jugroot C. Int J Hydrogen Energy. 2013;38:14595–14617. doi: 10.1016/j.ijhydene.2013.07.058. - DOI
-
- Satyapal S, Petrovic J, Read C, Thomas G, Ordaz G. Catal Today. 2007;120:246–256. doi: 10.1016/j.cattod.2006.09.022. - DOI
-
- Klerke A, Christensen CH, Nørskov JK, Vegge T. J Mater Chem. 2008;18:2304. doi: 10.1039/b720020j. - DOI
-
- Schüth F, Palkovits R, Schlögl R, Su DS. Energy Environ Sci. 2012;5:6278–6289. doi: 10.1039/C2EE02865D. - DOI
LinkOut - more resources
Full Text Sources
