Gut Microbiota Are Associated With Psychological Stress-Induced Defections in Intestinal and Blood-Brain Barriers
- PMID: 32010111
- PMCID: PMC6974438
- DOI: 10.3389/fmicb.2019.03067
Gut Microbiota Are Associated With Psychological Stress-Induced Defections in Intestinal and Blood-Brain Barriers
Abstract
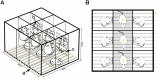
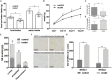
Altered gut microbiota has been identified during psychological stress, which causes severe health issues worldwide. The integrity of the intestinal barrier and blood-brain barrier regulates the process of bacterial translocation and can supply the nervous system with real-time information about the environment. However, the association of gut microbiota with psychological stress remains to be fully interpreted. In this study, we established a psychological stress model using an improved communication box and compared the expression of tight junction proteins in multiple regions of the intestinal (duodenum, jejunum, ileum) and blood-brain (amygdala, hippocampus) barriers between model and control rats. We also conducted fecal microbiota analysis using 16S rRNA gene sequencing. Expression levels of the stress-related indicators adrenocorticotropic hormone, NR3C1,2, and norepinephrine were increased in the model group compared to control group. Psychological stress reduced brain and intestinal levels of tight junction proteins, including claudin5, occludin, α-actin, and ZO-1. Microbiota analysis revealed elevated microbial diversity and fecal proportions of Intestinimonas, Catenisphaera, and Globicatella in the model group. Further analysis indicated a negative correlation of Allisonella and Odoribacter, as well as a positive correlation of norank_f__Peptococcaceae, Clostridium_sensu_stricto_1, and Coprococcus_2, with claudin5, occludin, α-actin, and ZO-1. Our use of a rodent model to explore the association between compromised intestinal and blood-brain barriers and altered fecal microbiota under psychological stress improves our understanding of the gut-brain axis. Here, cues converge to control basic developmental processes in the intestine and brain such as barrier function. This study provides new directions for investigating the pathogenesis of emotional disorders and the formulation of clinical treatment.
Keywords: blood–brain barrier; communication box; dysbiosis; gut microbiota; intestinal barrier; psychological stress; tight junction.
Copyright © 2020 Geng, Yang, Cheng, Zhang, Li, Liu, Li, Chen, Bao, Chen, Fei, Li, Hou, Lin, Liu, Zhang, Wang, Zhang, Wang, Wang and Zhang.
Figures




References
-
- Bailey M. T., Dowd S. E., Galley J. D., Hufnagle A. R., Allen R. G., Lyte M., et al. (2011). Exposure to a social stressor alters the structure of the intestinal microbiota: implications for stressor-induced immunomodulation. Brain Behav. Immun. 25 397–407. 10.1016/j.bbi.2010.10.023 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

