Human cardiac extracellular matrix-chitosan-gelatin composite scaffold and its endothelialization
- PMID: 32010293
- PMCID: PMC6966153
- DOI: 10.3892/etm.2019.8349
Human cardiac extracellular matrix-chitosan-gelatin composite scaffold and its endothelialization
Abstract
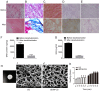
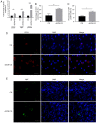
The present study developed a cardiac extracellular matrix-chitosan-gelatin (cECM-CG) composite scaffold that can be used as a tissue-engineered heart patch and investigated its endothelialization potential by incorporating CD34+ endothelial progenitor cells (EPCs). The cECM-CG composite scaffold was prepared by blending cardiac extracellular matrix (cECM) with biodegradable chitosan-gelatin (CG). The mixture was lyophilized using vacuum freeze-drying. CD34+ EPCs were isolated and seeded on the scaffolds, and then the endothelialization effect was subsequently investigated. Effects of the scaffolds on CD34+ EPCs survival and proliferation were evaluated by immunofluorescence staining and MTT assay. Cell differentiation into endothelial cells and the influence of the scaffolds on cell differentiation were investigated by reverse transcription-quantitative PCR (RT-qPCR), immunofluorescence staining and tube formation assay. The present results indicated that most cells were removed after decellularization, but the main extracellular matrix components were retained. Scanning electron microscopy imaging illustrated three-dimensional and porous scaffolds. The present results suggested the cECM-CG composite scaffold had a higher water absorption ability compared with the CG scaffold. Additionally, compared with the CG scaffold, the cECM-CG composite scaffold significantly increased cell survival and proliferation, which suggested its non-toxicity and biocompatibility. Furthermore, RT-qPCR, immunofluorescence and tube formation assay results indicated that CD34+ EPCs differentiated into endothelial cells, and the cECM-CG composite scaffold promoted this differentiation process. In conclusion, the present results indicated that the human cECM-CG composite scaffold generated in the present study was a highly porous, biodegradable three-dimensional scaffold which supported endothelialization of seeded CD34+ EPCs. The present results suggested that this cECM-CG composite scaffold may be a promising heart patch for use in heart tissue engineering for congenital heart disease.
Keywords: cardiac extracellular matrix; endothelial progenitor cells; endothelialization; heart patch; scaffold.
Copyright: © Lv et al.
Figures


References
-
- Benjamin EJ, Muntner P, Alonso A, Bittencourt MS, Callaway CW, Carson AP, Chamberlain AM, Chang AR, Cheng S, Das SR, et al. Heart disease and stroke statistics-2019 update: A report from the American heart association. Circulation. 2019;139:e56–e528. doi: 10.1161/CIR.0000000000000659. - DOI - PubMed
-
- Kochupura PV, Azeloglu EU, Kelly DJ, Doronin SV, Badylak SF, Krukenkamp IB, Cohen IS, Gaudette GR. Tissue-engineered myocardial patch derived from extracellular matrix provides regional mechanical function. Circulation. 2005;112(Suppl 9):I144–I149. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
