Aged garlic extract and its constituent, S-allyl-L-cysteine, induce the apoptosis of neuroblastoma cancer cells due to mitochondrial membrane depolarization
- PMID: 32010332
- PMCID: PMC6966145
- DOI: 10.3892/etm.2019.8383
Aged garlic extract and its constituent, S-allyl-L-cysteine, induce the apoptosis of neuroblastoma cancer cells due to mitochondrial membrane depolarization
Abstract
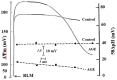
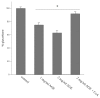
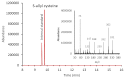
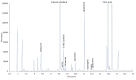
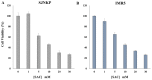
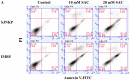
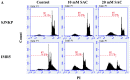
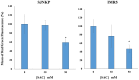
Aged garlic extract (AGE) has been demonstrated to have therapeutic properties in tumors; however its mechanisms of action have not yet been fully elucidated. A previous study revealed that AGE exerts an anti-proliferative effect on a panel of both sensitive [wild-type (WT)] and multidrug-resistant (MDR) human cancer cells. Following treatment of the cells with AGE, cytofluorimetric analysis revealed the occurrence of dose-dependent mitochondrial membrane depolarization (MMD). In this study, in order to further clarify the mechanisms of action of AGE, the effects of AGE on mitochondria isolated from rat liver mitochondria (RLM) were also examined. AGE induced an effect on the components of the electrochemical gradient (ΔµH +), mitochondrial membrane potential (ΔΨm) and mitochondrial electrochemical gradient (ΔpHm). The mitochondrial membrane dysfunctions of RLM induced by AGE, namely the decrease in both membrane potential and chemical gradient were associated with a higher oxidation of both the endogenous glutathione and pyridine nucleotide content. To confirm the anti-proliferative effects of AGE, experiments were performed on the human neuroblastoma (NB) cancer cells, SJ-N-KP and the MYCN-amplified IMR5 cells, using its derivative S-allyl-L-cysteine (SAC), with the aim of providing evidence of the anticancer activity of this compound and its possible molecular mechanism as regards the induction of cytotoxicity. Following treatment of the cells with SAC at 20 mM, cell viability was determined by MTT assay and apoptosis was detected by flow cytometry, using Annexin V-FITC labeling. The percentages of cells undergoing apoptosis was found to be 48.0% in the SJ-N-KP and 50.1% in the IMR5 cells. By cytofluorimetric analysis, it was suggested that the target of SAC are the mitochondria. Mitochondrial activity was examined by labeling the cells with the probe, 5,5',6,6'-tetrachloro-1,1',3,3'-tetraethylimidacarbocyanine iodide (JC-1). Following treatment with SAC at 50 mM, both NB cell lines exhibited a marked increase in MMD. On the whole, the findings of this study indicate that both natural products, AGE and SAC, cause cytotoxicity to tumor cells via the induction of mitochondrial permeability transition (MPT).
Keywords: S-allyl-L-cysteine; aged garlic extract; anti-proliferative activity; human neuroblastoma cancer cells; mitochondria; mitochondrial membrane depolarization.
Copyright: © Kanamori et al.
Figures



References
-
- Ohkubo S, Dalla Via L, Grancara S, Kanamori Y, García-Argáez AN, Canettieri G, Arcari P, Toninello A, Agostinelli E. The antioxidant, aged garlic extract, exerts cytotoxic effects on wild-type and multidrug-resistant human cancer cells by altering mitochondrial permeability. Int J Oncol. 2018;53:1257–1268. - PubMed
LinkOut - more resources
Full Text Sources
