The Role of RNA Binding Proteins for Local mRNA Translation: Implications in Neurological Disorders
- PMID: 32010708
- PMCID: PMC6974540
- DOI: 10.3389/fmolb.2019.00161
The Role of RNA Binding Proteins for Local mRNA Translation: Implications in Neurological Disorders
Abstract
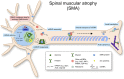
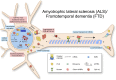
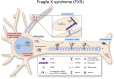
As neurons are one of the most highly polarized cells in our body, they require sophisticated cellular mechanisms to maintain protein homeostasis in their subcellular compartments such as axons and dendrites. When neuronal protein homeostasis is disturbed due to genetic mutations or deletions, this often results in degeneration of neurons leading to devastating outcome such as spinal muscular atrophy (SMA), amyotrophic lateral sclerosis (ALS), and fragile X syndrome (FXS). Ribonucleoprotein (RNP) complexes are macromolecular complexes composed of RNA binding proteins (RBPs) and their target RNAs. RBPs contain RNA binding domains and bind to RNA molecules via specific sequence motifs. RNP complexes have various functions in gene expression including messenger RNA (mRNA) trafficking, RNA processing and silencing. In neurons, RBPs deliver specific sets of mRNAs to subcellular compartments such as axons and dendrites to be locally translated. Mutations or deletions in genes coding for RNPs have been reported as causes for neurological disorders such as SMA, ALS, and FXS. As RBPs determine axonal or dendritic mRNA repertoires as well as proteomes by trafficking selective mRNAs and regulating local protein synthesis, they play a crucial role for neuronal function. In this review, we summarize the role of well-known RBPs, SMN, TDP-43, FUS, and FMRP, and review their function for local protein synthesis in neurons. Furthermore, we discuss their pathological contribution to the neurological disorders.
Keywords: ALS; FXS; RNA binding proteins; SMA; local protein synthesis; mRNA translation; neurodegeneration; ribonucleoproteins.
Copyright © 2020 Thelen and Kye.
Figures

Similar articles
-
Unraveling the Pathways to Neuronal Homeostasis and Disease: Mechanistic Insights into the Role of RNA-Binding Proteins and Associated Factors.Int J Mol Sci. 2018 Aug 3;19(8):2280. doi: 10.3390/ijms19082280. Int J Mol Sci. 2018. PMID: 30081499 Free PMC article. Review.
-
Altered mRNP granule dynamics in FTLD pathogenesis.J Neurochem. 2016 Aug;138 Suppl 1:112-33. doi: 10.1111/jnc.13601. Epub 2016 Jun 15. J Neurochem. 2016. PMID: 26938019 Review.
-
Phase separation and pathologic transitions of RNP condensates in neurons: implications for amyotrophic lateral sclerosis, frontotemporal dementia and other neurodegenerative disorders.Front Mol Neurosci. 2023 Sep 1;16:1242925. doi: 10.3389/fnmol.2023.1242925. eCollection 2023. Front Mol Neurosci. 2023. PMID: 37720552 Free PMC article. Review.
-
Drosophila as a Model for Assessing the Function of RNA-Binding Proteins during Neurogenesis and Neurological Disease.J Dev Biol. 2018 Aug 18;6(3):21. doi: 10.3390/jdb6030021. J Dev Biol. 2018. PMID: 30126171 Free PMC article. Review.
-
RNP granules in ALS and neurodegeneration: From multifunctional membraneless organelles to therapeutic opportunities.Int Rev Neurobiol. 2024;176:455-479. doi: 10.1016/bs.irn.2024.04.009. Epub 2024 May 14. Int Rev Neurobiol. 2024. PMID: 38802180 Review.
Cited by
-
Genome-Wide Mapping of RNA-Protein Associations via Sequencing.bioRxiv [Preprint]. 2024 Sep 4:2024.09.04.611288. doi: 10.1101/2024.09.04.611288. bioRxiv. 2024. PMID: 39282297 Free PMC article. Preprint.
-
RNA-Binding Proteins as Epigenetic Regulators of Brain Functions and Their Involvement in Neurodegeneration.Int J Mol Sci. 2022 Nov 23;23(23):14622. doi: 10.3390/ijms232314622. Int J Mol Sci. 2022. PMID: 36498959 Free PMC article. Review.
-
Illuminating RNA biology through imaging.Nat Cell Biol. 2022 Jun;24(6):815-824. doi: 10.1038/s41556-022-00933-9. Epub 2022 Jun 13. Nat Cell Biol. 2022. PMID: 35697782 Free PMC article. Review.
-
Impaired dynamic interaction of axonal endoplasmic reticulum and ribosomes contributes to defective stimulus-response in spinal muscular atrophy.Transl Neurodegener. 2022 Jun 2;11(1):31. doi: 10.1186/s40035-022-00304-2. Transl Neurodegener. 2022. PMID: 35650592 Free PMC article.
-
NKCC1, an Elusive Molecular Target in Brain Development: Making Sense of the Existing Data.Cells. 2020 Dec 4;9(12):2607. doi: 10.3390/cells9122607. Cells. 2020. PMID: 33291778 Free PMC article. Review.
References
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

