Prolonged cholestasis following endoscopic retrograde cholangiopancreatography, a rare complication of contrast agent induced liver injury: A case report and literature review
- PMID: 32011505
- PMCID: PMC7220498
- DOI: 10.1097/MD.0000000000018855
Prolonged cholestasis following endoscopic retrograde cholangiopancreatography, a rare complication of contrast agent induced liver injury: A case report and literature review
Abstract
Rationale: Prolonged cholestasis is a rare complication associated with endoscopic retrograde cholangiopancreatography (ERCP).
Patient concerns: A 68-year-old man who presented with worsening cholestasis after ERCP for the removal of a common bile duct stone.
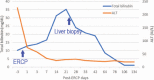
Diagnosis: Total bilirubin increased up to 35.2 mg/dL after the 21st day post-ERCP. A percutaneous liver biopsy was performed and drug-related cholestasis was suspected as occurring as a result of the contrast agent.
Interventions: Oral ursodeoxycholic acid and cholestyramine were prescribed to the patient.
Outcomes: By the 7th week post-ERCP, the patient's symptoms and markers of physiological health began to resolve. The bilirubin returned to normal levels on the 106th day post-ERCP. We reviewed the literature for studies of 9 patients with jaundice more than 30 days post-ERCP, the peak of total serum bilirubin occurred on 16th ± 7th days and the recovery followed after mean time of 54th ± 22th days.
Lessons: Although the cholestasis was prolonged, the outcome was favorable after medical therapy. There were no long-term consequences for the patient.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures

Similar articles
-
Prolonged cholestasis following successful removal of common bile duct stones: beware patients on estrogen therapy.World J Gastroenterol. 2007 Dec 14;13(46):6277-80. doi: 10.3748/wjg.v13.i46.6277. World J Gastroenterol. 2007. PMID: 18069774 Free PMC article.
-
Worsening cholestasis and possible cefuroxime-induced liver injury following "successful" therapeutic endoscopic retrograde cholangiopancreatography for a distal common bile duct stone: a case report.J Med Case Rep. 2016 Dec 21;10(1):371. doi: 10.1186/s13256-016-1123-0. J Med Case Rep. 2016. PMID: 28003028 Free PMC article.
-
An unusual case of prolonged post-endoscopic retrograde cholangiopancreatography jaundice.Hepatobiliary Pancreat Dis Int. 2016 Apr;15(2):220-2. doi: 10.1016/s1499-3872(15)60402-7. Hepatobiliary Pancreat Dis Int. 2016. PMID: 27020640
-
Bile duct disorders.Clin Liver Dis. 2003 May;7(2):295-309. doi: 10.1016/s1089-3261(03)00028-x. Clin Liver Dis. 2003. PMID: 12879985 Review.
-
Drug-induced vanishing bile duct syndrome: response to ursodiol.Am J Gastroenterol. 1996 Jul;91(7):1456-7. Am J Gastroenterol. 1996. PMID: 8678017 Review. No abstract available.
Cited by
-
A 107-day super-prolonged idiopathic cholestasis following endoscopic retrograde cholangiopancreatography.Gastroenterol Rep (Oxf). 2024 May 13;12:goae048. doi: 10.1093/gastro/goae048. eCollection 2024. Gastroenterol Rep (Oxf). 2024. PMID: 38745747 Free PMC article. No abstract available.
-
Hepatitis E Virus Infection Caused Elevation of Alanine Aminotransferase Levels in a Patient with Chronic Hepatitis B and Choledocholithiasis.Reports (MDPI). 2023 Nov 17;6(4):55. doi: 10.3390/reports6040055. Reports (MDPI). 2023. PMID: 40729178 Free PMC article.
References
-
- Adler DG, Baron TH, Davila RE, et al. American Society for Gastrointestinal Endoscopy. ASGE guideline: the role of ERCP in diseases of the biliary tract and the pancreas. Standards of Practice Committee of American Society for Gastrointestinal Endoscopy. Gastrointest Endosc 2005;62:1–8. - PubMed
-
- Anderson MA, Fisher L, Jain R, et al. ASGE Standards of Practice Committee. Complications of ERCP. Gastrointest Endosc 2012;75:467–73. - PubMed
-
- Dourakis SP, Mayroyannis C, Alexopoulou A, et al. Prolonged cholestatic jaundice after endoscopic retrograde cholangiography. Hepatogastroenterology 1997;44:677–80. - PubMed
-
- Lee HM, Bonis PAL, Kaplan MM. Persistent cholestatic jaundice after ERCP. Am J Gastroenterol 2006;101:204–5. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

